Study assesses proficiency gains when treating AF, SVT and PVCs without radiation
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/04d77f6f-fecc-4fd2-8db9-255ff9198e19/20-HVI-1915482-mapping-for-catheter-ablation-650x450-1_jpg)
20-HVI-1915482-mapping-for-catheter-ablation-650×450
The learning curve to gain proficiency in performing zero-fluoroscopy catheter ablations varies according to the type of ablation, concludes a recent study from Cleveland Clinic researchers published in the Journal of Interventional Cardiac Electrophysiology. The safety and efficacy of low- to no-fluoroscopy ablation has been established, but despite safety benefits from decreased radiation exposure, the practice has not been widely adopted.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“The biggest obstacle to widespread adoption of fluoroless ablation is operator experience and comfort,” says the study’s principal investigator, Roy Chung, MD, a Cleveland Clinic electrophysiologist. “It can be daunting to change the way you learned to do something, especially in procedures that involve complex anatomy.”
He and colleagues sought to quantify the operator learning curve necessary to perform fluoroless supraventricular (SVT), atrial fibrillation (AF) and premature ventricular contraction (PVC) ablations. They conducted a retrospective, single-center study of a single, junior faculty-level operator (Dr. Chung) pursuing zero fluoroscopy use.
“We thought that if we could identify the volume of cases necessary to achieve proficiency, we could help increase adoption of a practice that allows use of the least amount of radiation as reasonably achievable according to society guidelines,” says Dr. Chung. “We also were interested in pursuing this strategy to mitigate long-term lead wear, which has resulted in orthopaedic injuries. Individuals who benefit the most are our electrophysiology laboratory team members and anesthesia colleagues.”
The study included 167 patients who underwent ablation from 2016 to 2019. Seventy-seven patients (46%) underwent pulmonary vein isolation for AF, 64 patients (38%) had SVT ablations and 26 (16%) had PVC ablations. 3D electroanatomic mapping was used in all cases. The operator performed SVT ablations using standard atrial and ventricular stimulation. For patients with ventricular arrhythmias, only those with greater than 10% PVC burden received an ablation. All AF ablations were performed under general anesthesia.
Advertisement
“It quickly became clear that fluoroless transseptal access would be our rate-limiting factor,” says Dr. Chung. “An operator can certainly build confidence with this portion of the procedure, but it takes a bit more time.”
Key findings were as follows (see figure below):
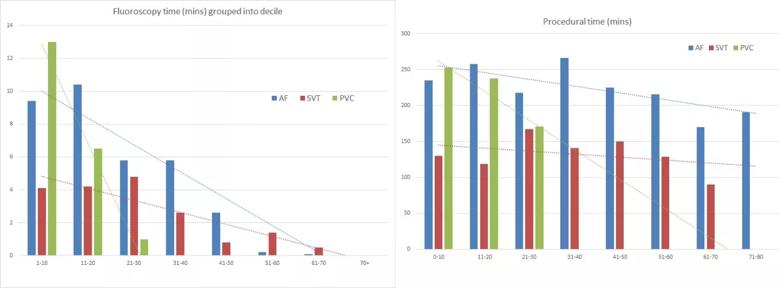
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/f30f6f23-cbf8-4c63-89a9-133e0d4eacde/Inset_png)
Figure. Fluoroscopy and procedural time stratified by consecutive cases of AF, SVT and PVC ablation. Reprinted by permission from Springer Nature, Journal of Interventional Cardiac Electrophysiology, “Operator learning curve and clinical outcomes of zero fluoroscopy catheter ablation of atrial fibrillation, supraventricular tachycardia, and ventricular arrhythmias,” Kochar A, et al. © 2020. Advance online publication 12 June 2020 (doi:10.1007/s10840-020-00798-8).
Advertisement
Previous studies have established the safety and efficacy of fluoroless catheter ablations; while this is a retrospective, single-site study, its findings offer a look at what it takes to learn a new way of performing ablation procedures.
“We’ve demonstrated that there appear to be distinct learning curves for each procedure, the steepest of which was for transseptal access using only intracardiac echocardiography guidance,” says Dr. Chung. “This was entirely out of our comfort zone at first but proved attainable and even seemed second nature by the end.”
He notes that zero fluoroscopy is not achievable in all circumstances, especially those involving patients with cardiac implantable electronic devices. But, he adds, as the use of three-dimensional mapping programs, contact-force-sensing catheters and intracardiac echo has become more widespread, the opportunity for reduced operator and patient radiation exposure is limited only by operator experience. “Now that we have an idea of what these learning curves look like, we can begin a more widespread adoption of these practices,” Dr. Chung says.
Study co-author Oussama Wazni, MD, concurs. “With a mindful approach, ablation with near-zero fluoroscopy is achievable while minimizing risk and optimizing operator experience,” says Dr. Wazni, Cleveland Clinic’s Section Head of Cardiac Electrophysiology and Pacing.
Advertisement
Advertisement
Large retrospective analysis may prompt prospective studies
How to talk about lifetime risk, treatment goals, Lp(a) testing, statin skepticism and more
A scannable recap of recent volumes and clinical metrics from Cleveland Clinic
Cleveland Clinic reports first U.S. series focused on use in this challenging setting
Large series confirms early and long-term survival advantages over partial pericardial resection
AVANT GUARD trial extends first-line role for ablation beyond paroxysmal atrial fibrillation
Maintain a high index of clinical suspicion and consider the underlying etiology
Protocol adoption at Cleveland Clinic sharply raised share of transferred patients getting timely PCI