Machine learning models shown to accurately separate lesions from normal brain, distinguish lesion subtypes
Success of epilepsy surgery is highly related to the identification and resection of epileptic lesions. Focal cortical dysplasia (FCD) is one of the most common lesions underlying medically intractable focal epilepsy. Conventional weighted MRI can be limited in detecting or characterizing subtle FCD, partly due to the lack of quantitative measurements for tissue properties. Now, for the first time, a study has demonstrated the value of a novel MR fingerprinting (MRF)-based radiomics method for providing objective, automated and accurate characterization of FCD lesions.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The study’s findings were reported in a Dec. 5 platform presentation at the American Epilepsy Society 2021 annual meeting by Joon Yu Choi, PhD, postdoctoral research fellow in the epilepsy imaging lab of Irene Wang, PhD, Research Director of Cleveland Clinic’s Charles Shor Epilepsy Center. The work was recognized with a Grass Foundation Young Investigator Award from the American Epilepsy Society.
MRF is a novel MR imaging method that can successfully address problems associated with conventional and advanced imaging modalities. MRF enables noninvasive quantification of multiple tissue properties simultaneously, through a novel approach to data acquisition, post-processing and visualization. The quantitative T1 and T2 measures that are generated from the MRF technique can be exquisitely sensitive to tissue alteration under pathological conditions such as epilepsy. NIH-funded research collaboration between Cleveland Clinic’s Dr. Wang and Dan Ma, PhD, Assistant Professor from Case Western Reserve University and one of the inventors of the MRF technology, has already led to the discovery of additional, clinically relevant findings in individuals with epilepsy (Ma et al., 2019).
“Here, to further utilize the exquisitely sensitive and quantitative brain maps, we developed a MRF-based radiomics feature extraction approach,” says Dr. Wang. “This approach can provide information for characterization of these subtle lesions that cannot be obtained using conventional MRI.”
Between 2019 and 2021, 29 patients with medically intractable focal epilepsy and FCD, as well as 49 sex- and-age matched controls, were recruited at Cleveland Clinic’s Charles Shor Epilepsy Center. All underwent a 3D whole-brain MRF scan using a 3T MRI scanner. High-resolution MRF T1, T2, gray matter, white matter and T1w maps were generated based on direct matching to a predefined dictionary (Figure).
Advertisement
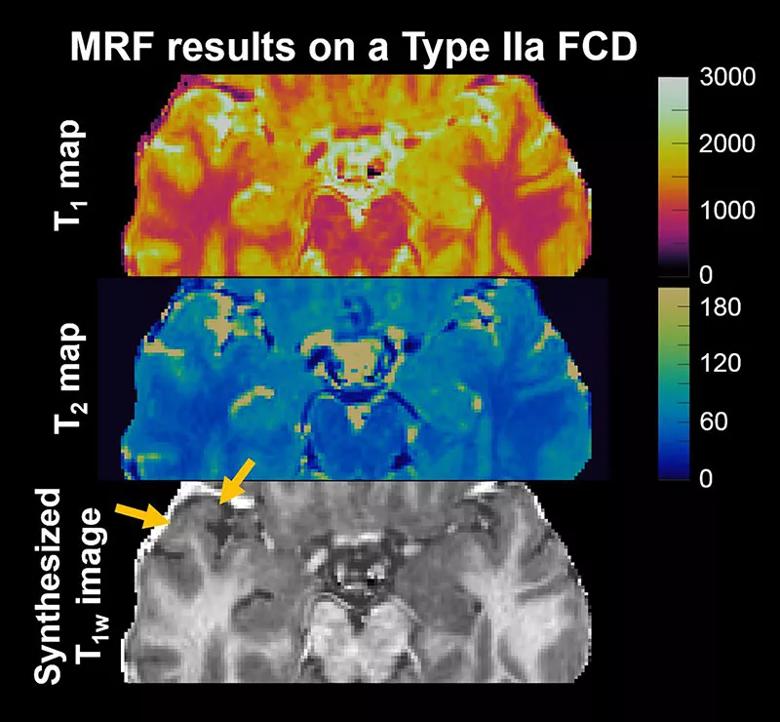
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/0f4fefc4-d7ef-405f-b44c-2c335f091d32/21-NEU-2536040-CQD-Inset-800x740-1_jpg)
Figure. MRF images from a patient with histopathologically confirmed FCD type IIa that was not detected by conventional MRI. On the T1 map, the anterior aspect of the right superior temporal gyrus shows higher signal intensities compared with the contralateral side (upper panel). In addition, the T1 map in the lesional region shows a “speckled” appearance, which likely comes from higher variations within the lesion (upper panel). The synthesized T1w image shows blurring of the gray matter/white matter junction (lower panel), a typical MRI characteristic for FCD.
3D lesion ROIs were manually created on the MRF images. Images from patients and healthy controls were normalized to a common space and compared with each other within the 3D ROIs. A total of 192 radiomics features in the ROIs were extracted from MRF T1, T2, gray matter and white matter maps. Logic regression machine learning models were built to separate FCD from normal tissue, and to distinguish FCD type I from FCD type II. Datasets were divided into a training set (80%) and a testing set (20%). After features with the highest average accuracy were selected in the training set, the models with these optimal features were validated in the testing set. Performance metrics were averaged from 10 randomly shuffled datasets to minimize bias.
Key findings included the following:
Advertisement
“The novelty and strength of our approach is that MRF is multiparametric and quantitative in nature,” Dr. Wang observes. “Combining this powerful imaging technique with radiomics methods that provide quantitative, computer-extracted imaging biomarkers will likely lead to generation of information that is invisible to conventional MRI and highly relevant to the pathology. This is exactly what we are seeing with the current study.”
Cleveland Clinic experience demonstrates the potential of MRF to detect more subtle lesions that were missed on conventional MRI, according to a previously published study by this research team (Ma et al., 2019).
“Because the MRF radiomics machine learning approach is automated and not dependent on users, it can potentially be used as an objective and individualized tool to aid the presurgical evaluation of patients with epilepsy,” Dr. Wang says.
She adds that her Cleveland Clinic team will continue to combine the novel MRF technique with post-processing analyses and machine learning/deep learning algorithms. “This combination of innovations will allow for unleashing the power of MRF for epilepsy clinical applications,” she concludes.
Ma D, Jones SE, Deshmane A, Sakaie K, Pierre EY, Larvie M, McGivney D, Blümcke I, Krishnan B, Lowe M, Gulani V, Najm I, Griswold MA, Wang ZI. Development of high-resolution 3D MR fingerprinting for detection and characterization of epileptic lesions. J Magn Reson Imaging. 2019;49(5):1333-1346.
Advertisement
Advertisement

Distinct MRI signature includes lesions beyond the corpus callosum, features predictive of vision and hearing loss

An argument for clarifying the nomenclature

An expert talks through the benefits, limits and unresolved questions of an evolving technology

Recommendations on identifying and managing neurodevelopmental and related challenges

Phase 2 trials investigate sitagliptin and methimazole as adjuvant therapies

Aim is for use with clinician oversight to make screening safer and more efficient

Rapid innovation is shaping the deep brain stimulation landscape

Study shows short-term behavioral training can yield objective and subjective gains