Advertisement
Cleveland Clinic findings challenge conclusions of ARUBA trial
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Surgical treatment of unruptured brain arteriovenous malformation (ubAVM) is associated with a lower risk of stroke or death than a watch-and-wait approach, according to a new retrospective analysis of a large series of cases at Cleveland Clinic. These results add to the controversy generated with the 2014 publication of the Randomized Trial of Unruptured Brain Arteriovenous Malformations (ARUBA),1 which came to the opposite conclusion. Our retrospective analysis of 105 patients between 2001 and 2014 who would have qualified for ARUBA was recently published in Neurosurgery.2
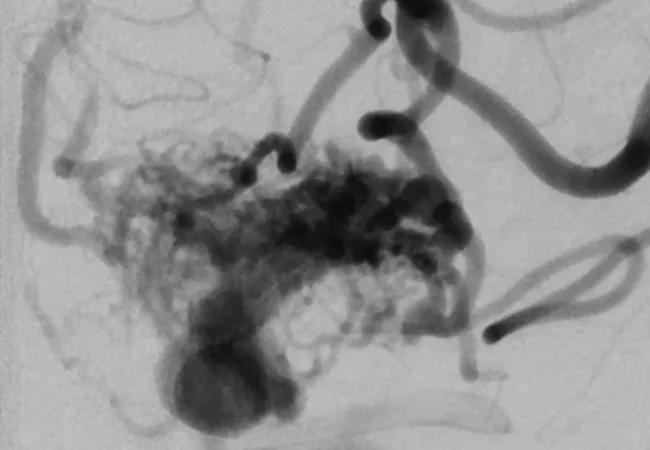
Optimal management of ubAVMs has been a topic of debate since brain imaging made possible the diagnosis of this often asymptomatic or mildly symptomatic condition (an example angiogram of such an AVM is shown at the top of this post). A no-treatment strategy entails a 2 percent to 5 percent annual risk of rupture. While this may seem low, over many years the cumulative chances become daunting. And the outcomes can be devastating: About one-third of patients with a bleed end up in a nursing home, and 10 percent die. But treatment is also not without risk.
ARUBA, conducted at 39 clinical sites in nine countries, randomized patients to either interventional therapy (neurosurgery, embolization or stereotactic radiosurgery [Gamma Knife®], either in combination or alone, as determined by the treating physician) or medical management (symptomatic treatment with medications, as needed). The study, which started in 2007, was ended prematurely in 2013 because of superior outcomes in the medical management group. At that point, the study had randomized 223 patients (114 to intervention and 109 to medical management) and had a mean follow-up of 33.3 months. The primary end point, either stroke or death, had occurred in 10.1 percent of the medical management group versus 30.7 percent of the intervention group.
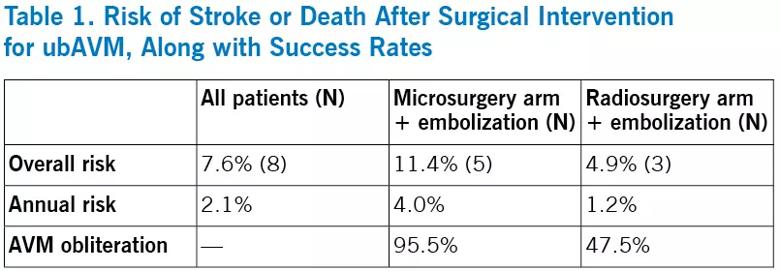
Of the 105 cases in the Cleveland Clinic database that were ARUBA-eligible, 44 patients had undergone microsurgery and 61 patients had undergone stereotactic radiosurgery as their final treatment. Mean follow-up was 43 months (range, 4-136 months). A total of 8 patients (7.6 percent) had a stroke or died, as detailed in the table below.
This new Cleveland Clinic study2 joins several other retrospective analyses published after ARUBA in finding substantially lower overall risk of stroke or death in the treatment arm than the 30.7 percent reported in ARUBA.3-6 Most importantly, overall risk was lower than the natural disease course, justifying a strategy of surgical intervention for ubAVM.
Multiple issues have been identified that may account for the different conclusions reached by ARUBA and the other studies.
Treatment choices differed. ARUBA treated 26 percent of patients with endovascular embolization alone, a high rate that may partly explain the elevated rate of stroke or death relative to the subsequent studies. Because of the likelihood of complications from attempting to obliterate an AVM by embolization alone, many institutions, including Cleveland Clinic, reserve its use mainly to reduce AVM nidus size before surgery or stereotactic radiosurgery. With safer embolysates now available, the role of embolization in combined procedures is undergoing a further shift.
Cleveland Clinic uses the following rules of thumb to choose interventions for a patient with ubAVM:
Advertisement
Short follow-up. Because most deaths from surgical intervention happen soon after the procedure and the risk of an untreated ubAVM is generally continuous over the years, a comparison of the risks within the first few years is likelier to favor no intervention.
Treatment of many patients outside the trial. ARUBA was designed such that not all patients who qualified for the trial were randomized, allowing surgeons to elect to operate on patients whose AVMs were deemed ideal for surgery. This ability to treat patients outside the trial could have introduced bias by steering disproportionate numbers of patients with complex or difficult-to-treat AVMs into the trial. This would not have been favorable for the trial’s interventional arm.
Another randomized clinical trial would be ideal, using a more modern algorithm for treatment than was used in ARUBA. But because of the high expense, this is unlikely to happen soon. A second choice would be a prospective study conducted at multiple institutions to follow outcomes of different treatment strategies. Finally, a meta-analysis of the various retrospective studies that have followed ARUBA, including this Cleveland Clinic cohort, would be useful.
We believe in providing our ubAVM patients with as much information as possible so that they can fully enter into the decision process for their treatment. They often have strong preferences based on the degree of risk they are comfortable with.
Our inclination to intervene stems from witnessing young, healthy patients meet with poor outcomes from a wait-and-see approach. Our data strongly argue that we have the tools to provide reasonable and safe strategies to improve outcomes for most patients.
Advertisement
Dr. Bain is a neurosurgeon in Cleveland Clinic Neurological Institute’s Cerebrovascular Center. He was primary investigator of the retrospective analysis profiled above.
Advertisement
Advertisement
Real-world claims data and tissue culture studies set the stage for randomized clinical testing
New grant-funded investigation illustrates impact and reach of Cleveland Clinic Brain Study
How the new longitudinal investigation could become the Framingham Heart Study of brain health
Pilot findings show good patient acceptance and safety, early hints of efficacy

Study finds high prevalence of symptoms, willingness to seek treatment
Panel outlines research priorities around a promising alternative to imaging markers
Suspected factors include antithrombotic intensity, time on device, presence of thrombocytopenia
Preclinical studies will assess whether method developed for stroke recovery curbs deficits after brain injury