A public health tragedy with persistent pathophysiological and therapeutic challenges

Written by Philippe Haouzi, MD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
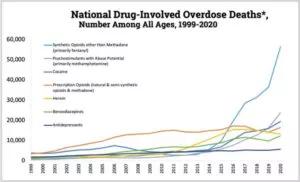
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/fd471eb0-a328-4c65-b791-a3e4a8d744f3/22-PUL-3270759-CQD-The-respiratory-toxicity-of-opioids-in-2022-inset-300x182_jpg)
Figure 1: Unintentional deaths by drug poisoning in the US since 1999. Note that death by fentanyl (blue line) has represented the most deaths by drug poisoning in the country over the last few years. (Developed by the Centers for Disease Control and Prevention, National Center for Health Statistics, released 12/2021. Use of the material does not imply endorsement by CDC, ATSDR, HHS or the United States Government of Cleveland Clinic.)
Opioid-induced acute ventilatory depression is responsible for the life-threatening toxicity of opioids, as it can lead to a terminal hypoxic/anoxic cardiac arrest, by pulseless electrical activity or ventricular arrhythmia.1 For multifarious but not fully understood reasons, there has been a dramatic increase in deaths by opioid overdose over the last decade in the US. The data recently released by the CDC are extremely concerning. Among the ~ 100,000 annually identified unintentional overdose deaths in the U.S (involving any drugs or medications), about 70% are currently related to the use of opioids, such as fentanyl (Figure 1).2 Opioid-induced breathing depression can affect not only addicted individuals using intravenous injections, but also various subsets of patients. For instance, patients with obesity hypoventilation, sleep-disordered breathing, neuromuscular deficit, chronic restrictive or obstructive lung diseases, and patients in a post-operative setting, have very high risks of opioid-induced ventilatory depression, with sometimes poor outcomes.3,4 How to better understand and better treat opioid-induced ventilatory depression in these different populations is at the center of the new NIH-funded research program recently developed by the Department of Pulmonary Medicine at Cleveland Clinic. This program is addressing some of the following outstanding questions:
Advertisement
Recovering from an acute depression in breathing produced by potent opioid agonists, such as fentanyl, depends on multiple factors critical for survival. For instance, when severe levels of hypoxemia are reached (PaO2 < 20 mmHg), spontaneous recovery from an acute opioid intoxication is not possible and naloxone becomes totally ineffective.5 This observation implies that ventilatory support must be provided before naloxone administration in any victims of opioid overdose found apneic. Also, mu-opioid receptor desensitization, produced by a mechanism referred to as the “GRK2/b-arr2” pathway, could be essential for breathing recovery. 6 This pathway allows for a rapid recovery, within minutes, from the depressive effects of an opioid agonist on neurons. Drugs capable of increasing specifically this activity would represent an interesting approach in patients with a high risk of breathing depression.6 Association with other medications can also alter this desensitizing pathway. For example, the SSRI, paroxetine, has been shown to depress the “GRK2/b-arr2” activity; it should be avoided, until proven otherwise, in patients taking opioids. Finally, opioid-induced muscle rigidity adds potentially lethal consequences to the depression of the drive to breathe.7-9 Opioid-induced muscle rigidity produces tonic contractions of the chest and abdominal muscles, typically associated with a glottic closure in turn impeding breathing recovery. This effect literally counteracts breathing movements for prolonged periods of time.8-9 The antidotal properties of Kappa and central alpha 2 agonists on this curious, yet life-threatening, breathing side effect of opioids are currently being investigated in pre-clinical studies.7
Advertisement
Whenever a life-threatening ventilatory depression is present, the use of large doses of naloxone along with cardiopulmonary resuscitation maneuvers, if needed, remains the standard of care.10 The risk of withdrawal or of suppressing analgesia does not outweigh the need to restore breathing as soon as possible. However, in patients who are not presenting an immediate lethal risk, a correction of opioid-induced respiratory depression only (without reversing the other effects of opioids) would be an appropriate approach.4 This strategy is certainly clinically relevant in a post-operative care setting when sparing the analgesic effects of opioids is crucial. Also, addicted patients, who can present violent symptoms of acute withdrawal when receiving a large dose of naloxone, would benefit from a reversal of hypoventilation only, if no immediate life-threatening risk is present. There is no currently accepted treatment that can be offered to only overcome the potent depressive effects of opioids on breathing, despite many attempts to develop new therapeutic agents (from monoclonal antibodies to ventilatory stimulants) as well as new paradigms using already FDA-approved antidotes.
The recent changes aimed at stricter regulations of opioid use and easier access to naloxone, did not have the impact that many expected on death by opioid overdose. Yet, efforts must be pursued to better understand the mechanisms of recovery from opioid-induced breathing depression, offer successful prevention strategies and develop alternate countermeasures to restore breathing.
Advertisement
REFERENCES
Advertisement
Advertisement

Case study illustrates the potential of a dual-subspecialist approach
New tools and protocols to improve care

Endoscopic balloon dilation during pregnancy helps optimize outcomes

Surgery is typically the only option for the most severe cases, but a minimally invasive procedure is reducing morbidity and recovery time for patients
Cleveland Clinic pulmonologists share a framework for how to implement effective clinical protocols to standardize evaluation and management of complex acute respiratory distress syndrome

Recent breakthroughs have brought attention to a previously overlooked condition

Expert urges action to mitigate the tragedy of fatal overdose

Findings show profound muscle loss variance between men and women