Exploring the responses to medications and other supportive therapies
Written by Vidula Vachharajani, MD, Matt Siuba, DO, and Siddharth Dugar, MD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Sepsis, a dysregulated immune response to infection, causes life-threatening organ dysfunction in nearly 50 million people each year and leads to 11 million deaths worldwide. The cardiovascular system is at the center of sepsis-related organ dysfunction.
Immune response in sepsis transitions from early hyperinflammation to a late persistent inflammation and immunosuppression (PII) phase. The PII phase of sepsis is particularly deadly since the majority of sepsis-related deaths occur during this phase (Figure 1). Emerging evidence suggests that epigenetic and metabolic factors are responsible for the transition from hyperinflammation to PII.
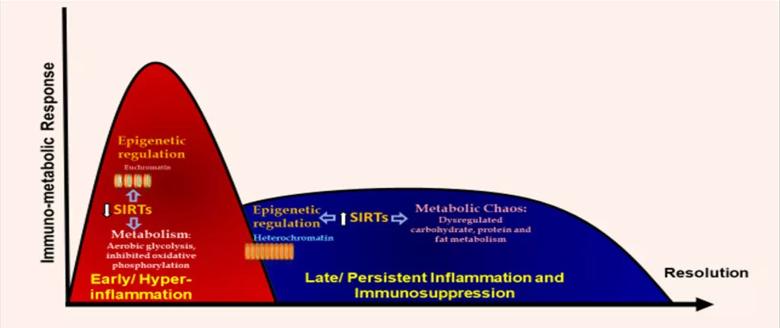
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/e2a58298-fde3-47a5-9665-540758faac1f/23-PUL-4178625-CQD-DM-Unravelling-mechanisms-sepsis-Inset-1-1-1024x431_jpg)
Figure 1. Early (Hyperinflammatory) and late phase of Sepsis.
The laboratory of Vidula Vachharajani, MD, studies how immune cell metabolism affects immune response in sepsis with two common comorbidities: obesity and alcohol use disorder. Both comorbidities influence the nutritional status of a cell/organism. Specifically, the laboratory focuses on the role of sirtuins in the transition of immune response from hyperinflammation to PII. The sirtuins are known as nutrient sensors and epigenetic modulators.
Multiple publications from the laboratory have shown that sirtuin 1 (SIRT1) and sirtuin 2 (SIRT2) expressions decrease during hyperinflammation and show sustained increase during the PII phase. The laboratory is currently funded by the National Institutes of Health (NIH) R01 mechanism. One of its focuses is on the role of sirtuin activation in alcohol with sepsis phases and immune-metabolic responses responsible for ethanol-induced immune repression. Similarly, the laboratory continues to study the role of sirtuins in influencing the outcomes of sepsis patients via NIH R35 funding.
Advertisement
The lessons learned in the Vachharajani laboratory are intimately tied to work being done by two clinician-scientists working with critically ill patients at Cleveland Clinic. The work from Siddharth Dugar, MD, and Matt Siuba, DO, focuses on studying the hemodynamic changes associated with sepsis and septic shock in critically ill patients.
More specifically, Dr. Dugar is studying the relationship of left ventricle (LV) systolic function and mortality in sepsis and septic shock (Figure 2). His preliminary work has provided a novel way to evaluate LV systolic function in patients with septic shock. Dr. Dugar’s initial studies have shown that in patients with septic shock, higher mortality is observed both in patients with impaired function and in those with hyperdynamic heart function. This changes the paradigm of how cardiac function is studied in these populations.
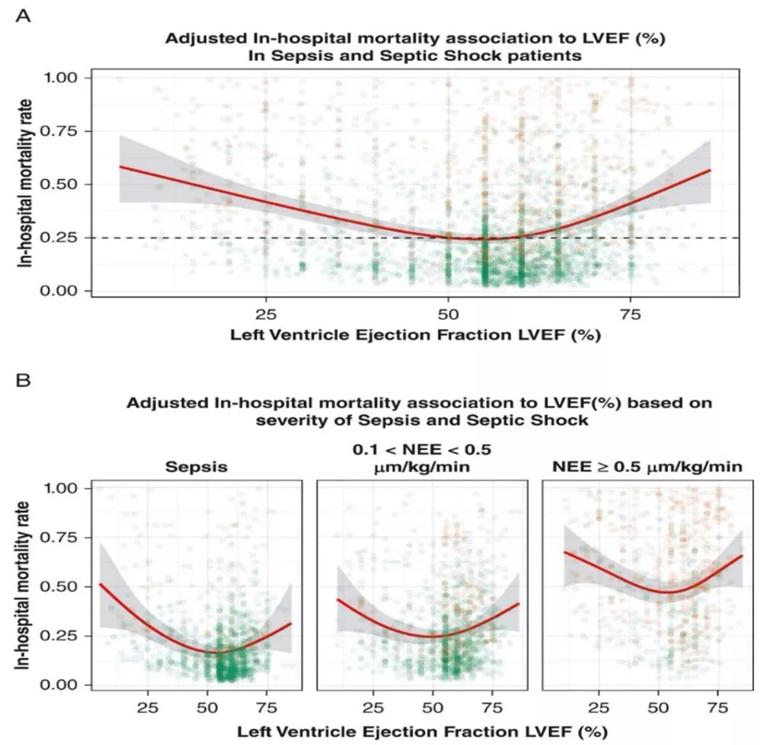
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/ef934884-038f-4480-b65d-241e53ffe247/23-PUL-4178625-CQD-DM-Unravelling-mechanisms-sepsis-Inset-2-1-1024x978_jpg)
Figure 2. Hospital mortality according to LVEF (%) in all critically ill patients with sepsis (above) and in subgroup of sepsis, septic shock requiring < 0.5 μg/kg/min of norepinephrine dose equivalent and septic shock requiring ≥ 0.5 μg/kg/min of NEE equivalent (below). The relationship of left ventricle systolic function to mortality is U-shaped in each subgroup based on sepsis severity. (LVEF = left ventricular ejection fraction; NEE = norepinephrine equivalent
dose.)
(Reprinted from Chest, 163/6, Dugar S, Sato R, Chawla S, et al. Is Left Ventricular Systolic Dysfunction Associated With Increased Mortality Among
Patients With Sepsis and Septic Shock?, 1437-1447, 2023, with permission from Elsevier.)
Advertisement
Similarly, Dr. Siuba has recently received funding to study the impact of changes in the right ventricle in patients with shock and respiratory failure. His preliminary work is groundbreaking as he studies the impact of changes to right ventricular function in critically ill patients by studying pressure-volume relationships and how these changes impact outcomes in these populations. His future research will be focused on acute hemodynamic changes to the right ventricle in clinical syndromes such as sepsis, septic shock and acute respiratory distress syndrome, especially under mechanical ventilation.
This targeted hemodynamic and immunologic profiling can help us identify specific phenotypes and identify differential hemodynamic responses to therapies. This groundbreaking work from clinician-scientists at Cleveland Clinic is breaking the silos in phenotyping patients with sepsis and septic shock. A targeted effort to identify and study these immunologic and hemodynamic profiles is the foundation of understanding the differential responses to medications and other supportive therapies. It will be instrumental in identifying the heterogeneity of treatment effects in specific populations with sepsis and septic shock.
Advertisement
Advertisement

Takeaways from the most recent annual meeting centered around clinical advances, AI integration and professional development

Cleveland Clinic research emphasizes taking a holistic and individualized approach to care of septic shock

Transvalvular LVAD support transforms emergency surgery into urgent procedure

Aortic valve replacement is best option for lowering mortality in this high-risk population
Preoperative Impella 5.5 placement can provide a critical safety net for high-risk patients
Young man saved multiple times by rapid collaborative response

Recent breakthroughs have brought attention to a previously overlooked condition

A review of treatment options for patients who may not qualify for surgery