New technology creates patient-specific silicone stent
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/44c1a0eb-90ef-4df6-b0b5-bb8d146b1f23/GettyImages-1389278592-scaled-jpg)
Lungs
By Thomas Gildea, MD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
A 58-year-old man with a history of lung cancer successfully treated with surgery, radiation therapy and chemotherapy 13 years ago presented to Cleveland Clinic for evaluation of severe dyspnea.
CT scan of the chest showed stricture of the distal trachea and right main bronchus related to his prior cancer treatment. After standard bronchoscopic techniques (i.e., electrosurgery cuts, balloon dilation) proved ineffective in ameliorating his shortness of breath, a decision was made to place a Y stent.
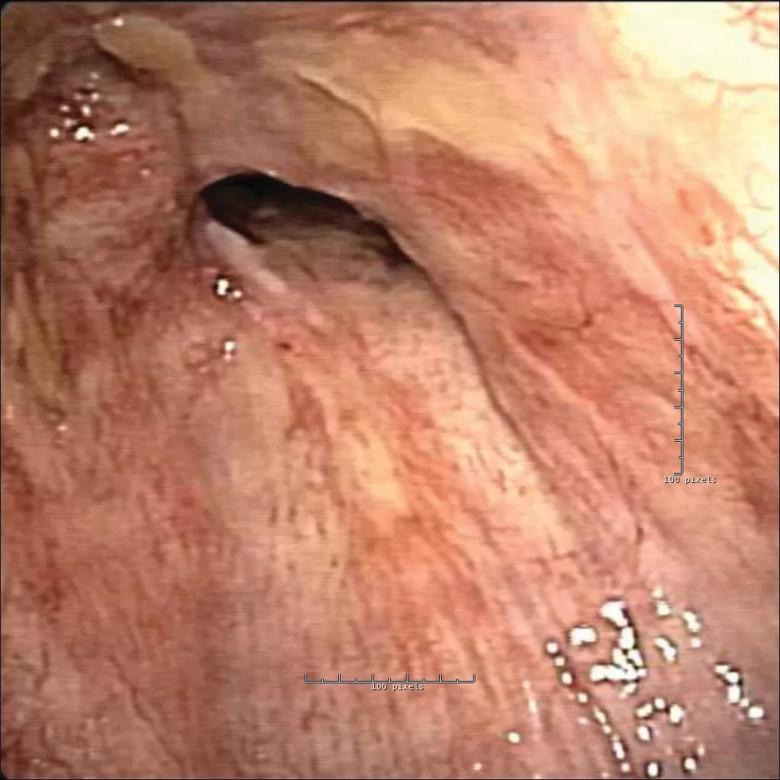
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/4b5a9fe8-bf48-4f6c-a2f6-f3c614add34d/Figure-1A_jpg)

Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/19668c9e-a36a-4cc4-8ca1-5e2f1278ddc1/18-PUL-4959-Gildea-Image-1B-650pxl-width_jpg)
Because the right and left bronchus were different sizes and the angle to the right bronchus was distorted from radiation, a standard Y stent would not fit. Modifications were needed to customize the stent for the abnormal anatomy. This was accomplished by sewing together multiple stents at various angles to effectively open the airway.
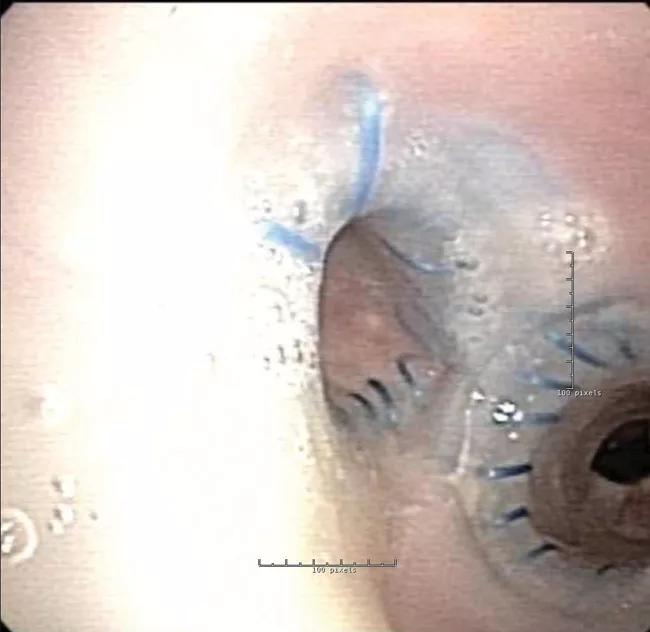
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/64abe536-4793-4984-9796-791cf6c690ae/18-PUL-4959-Gildea-Image-1C-650pxl-width_jpg)
However, due to the stent geometry and underlying disease, granulation tissue developed and occluded the airway.
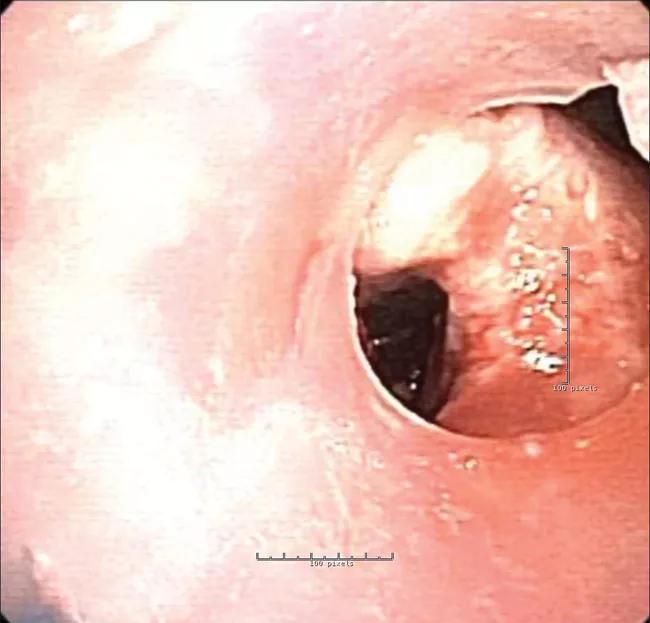
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/38514977-9770-4a6a-80e4-ba9ec479104b/18-PUL-4959-Gildea-Image-1D-650pxl-width_jpg)
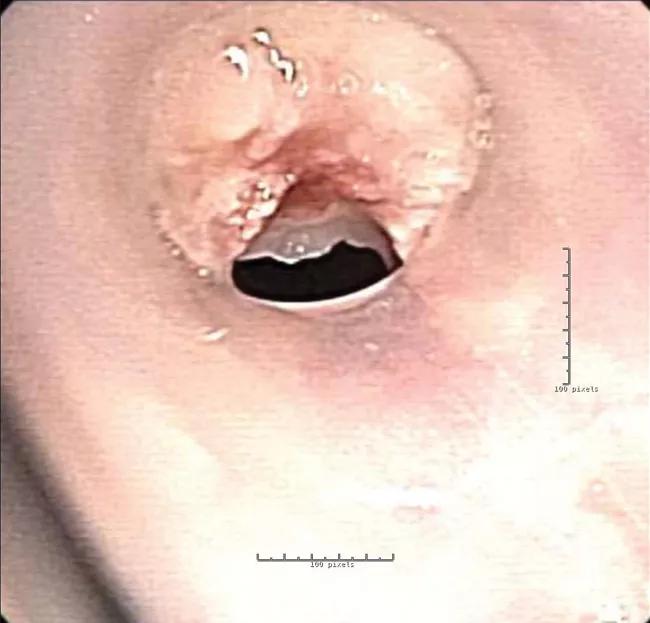
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/2d228e68-29aa-4009-931e-0620abf30b83/18-PUL-4959-Gildea-Image-1E-650pxl-width_jpg)
Procedures to clean or readjust the stent became necessary every four to six weeks, then eventually every two weeks. In response, the interventional pulmonology staff contacted the FDA to request compassionate use for a patient-specific stent made using 3D-printing technology. A stent designed using the native airway as a base model would prevent pressure points (to avoid granulation) and improve airflow.
While waiting for FDA approval, the patient was admitted to the intensive care unit with acute respiratory failure due to mucus plugging. He underwent emergent bronchoscopy to clean out the existing stents. Given the urgency of the situation, the interventional pulmonary team, with approval of Cleveland Clinic’s institutional review board, alerted the FDA that they were proceeding with plans to expeditiously develop a patient-specific Y stent.
Advertisement
A software engineer uploaded CT images of the airway and converted them into a 3D model using a web-based program.

Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/4ac825f6-7b4d-4382-8f71-74581260c35b/18-PUL-4959-Gildea-Hero-Image-650x450pxl_jpg)
Then a physician submitted a prescription for a patient-specific stent to be built to treat the diseased areas. Software engineers converted the prescription into a 3D printed mold used to form the stent with medical-grade silicone. After production, the stent was sterilized before implantation.
In the bronchoscopy suite, the patient was placed under general anesthesia, and rigid bronchoscopy was performed to remove the old stents. The new patient-specific stent was loaded and deployed with standard rigid instrumentation and easily positioned into the planned treatment area.
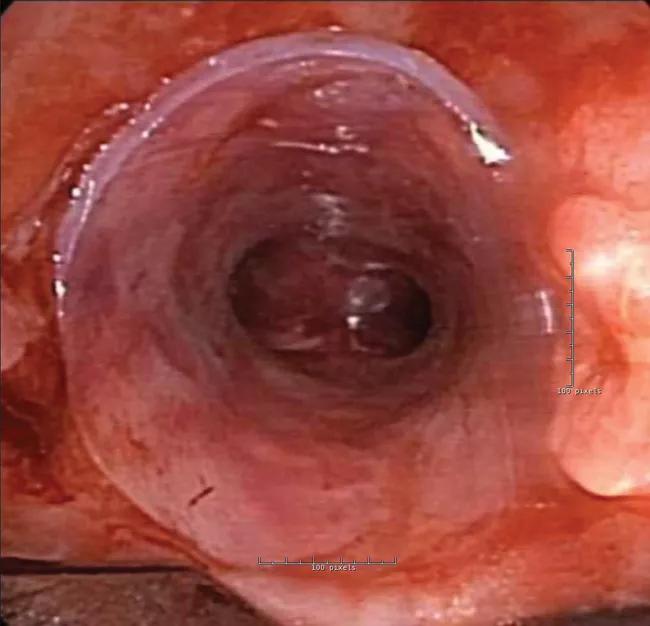
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/204fca7b-df4d-4e27-9288-a733dedeb7c9/18-PUL-4959-Gildea-Image-2B-650pxl-width_jpg)
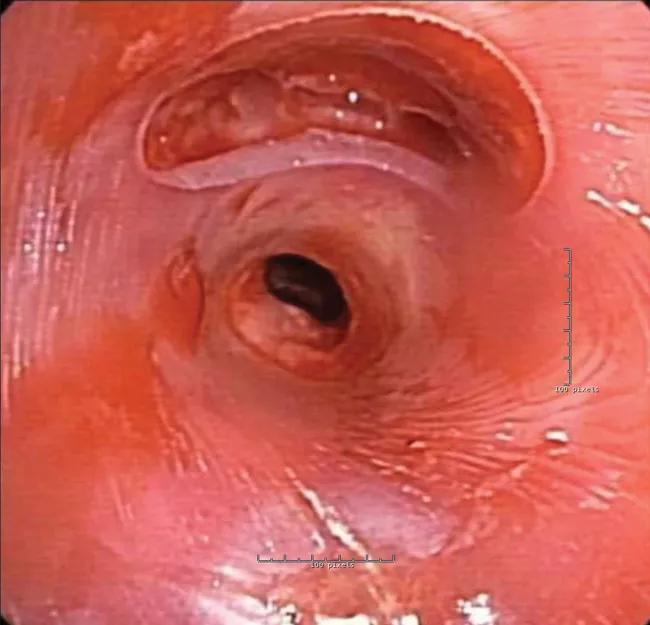
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/cdf33bf8-2469-40fb-a6d6-f81e56ce2ca3/18-PUL-4959-Gildea-Image-2C-650pxl-width_jpg)
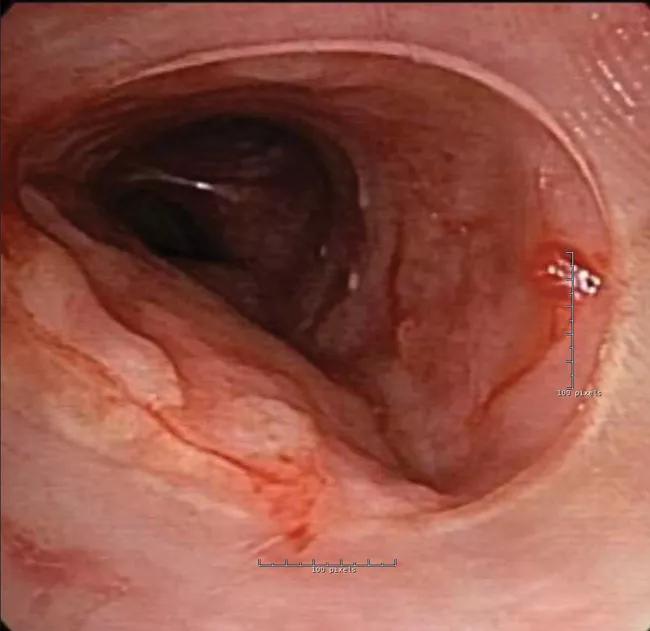
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/623951e1-5639-476f-9d4f-b2272829ffa3/18-PUL-4959-Gildea-Image-2D-650pxl-width_jpg)
The patient was observed in the intensive care unit for 48 hours and subsequently discharged. He has recovered fully and continues standard follow-up, including bronchoscopic procedures every several months to assess and clean the stent.
He has not required any hospitalizations or emergency procedures since placement of the stent. He has experienced marked improvement in his quality of life, sleeping in bed instead of in a recliner and returning to work after three years on disability.
Patient-specific stents made with 3D technology are currently under FDA review and approved only for compassionate use on a case-by-case basis. Cleveland Clinic pioneered this advanced technology and has provided it for qualified patients since 2016.
Advertisement
Advertisement
The Center for Environment, Place and Health Research connects clinicians with mapping tools and other real-world data resources to help guide medical insights
Exploring the true prevalence of AATD among Americans and why so many remain undetected
Rapid growth and collaboration have advanced the Lung Nodule Program, but patient engagement barriers persist
A physician's perspective on confirmation imaging, diagnostic confidence and choosing the right robotic platform for patients
A look into recent developments in the diagnosis of LAM and its clinical presentation
Takeaways from the most recent annual meeting centered around clinical advances, AI integration and professional development
Recent breakthroughs have brought attention to a previously overlooked condition
A review of treatment options for patients who may not qualify for surgery