Sophisticated in vivo modeling can enhance device development and therapy testing

Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/b2ead052-2b5c-4836-91e8-c07b47e0bd5f/22-HVI-3200902-CQD-650x450-1_jpg)
22-HVI-3200902 – CQD 650×450
A soft robotic device, fitted around the ascending aorta in a living swine, enables high-fidelity simulation of hemodynamics and biomechanics of aortic stenosis and congenital aortic valve anomalies. The cuff-shaped sleeve — which can be personalized based on a patient’s MRI or CT data — has the potential to serve as a model for testing therapies and guiding clinical management. The system was created by a team of biomedical engineers, imaging experts and cardiovascular clinicians and is described in Nature Biomedical Engineering (2022;6[10]:1134-1147).
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“Our highly tunable and customizable sleeve realistically recapitulates the hemodynamics of calcific aortic stenosis and congenital valve disease,” says the article’s corresponding author, Christopher Nguyen, PhD, Director of MRI Research in Cleveland Clinic’s Heart, Vascular & Thoracic Institute. “The ability to closely recreate patient-specific conditions paves the way toward developing personalized approaches to better understand and treat aortic stenosis.”
So-called soft robotics — i.e., technologies using pliable materials with qualities similar to biological tissues — can be designed to mimic biomechanical function and complex motion dynamics of various organs and systems. They are increasingly being developed as models of human physiology and disease. Dr. Nguyen previously led a team to create a soft robotic heart model that simulates contractile motion with the potential for use in preclinical testing of cardiac devices (Sci Robot. 2020;5[38]:eaay9106).
However, existing preclinical models of aortic stenosis are simplistic, often consisting of a rubber tube with sutures tightened around it to simulate the constriction of vessel stenosis. Such systems do not account for complex flow patterns caused by different configurations of the aortic valve, pulsatile valvular movement and other natural mechanisms of physiological change that occur within a living being.
This current study reports on the development of a more advanced in vivo model that functions in a living swine with information from CT, 4D MRI and other tools to more accurately recreate the hemodynamics of aortic stenosis resulting from calcific aortic valve or congenital valve disease.
Advertisement
The sleeve, which fits as a band around the ascending aorta, consists of two thermoplastic polyurethane sheets heat-sealed together, formed with a 3D printer to contain three empty pockets. “We realized that achieving biomimicry of the native aortic valve anatomy through our sleeve design was critical to recapitulate aortic stenosis with high fidelity,” says lead author Luca Rosalia, a graduate student in the Health Sciences and Technology program of Harvard and the Massachusetts Institute of Technology (MIT).
Each pocket of the sleeve is expandable and customizable by pumping in water or air. When wrapped around the swine aorta with the pockets facing inward, the device mimics the trifold shape of an aortic valve (or the shape of a unicommissural or bicommissural valve by inflating only one or two of the pockets). The pockets are each connected to an actuation line to enable varying volume and pressure parameters, allowing for the recreation of realistic, individualized hemodynamics. “Being able to re-create congenital defects and to finely control associated hemodynamics is an incredible leap forward in the development of high-fidelity test beds of cardiovascular disease,” notes co-author Rosalia.
“Beyond this exemplar, we envision using tunable soft robotic tools to recapitulate the mechanics and hemodynamics of various cardiovascular diseases in the lab (in vitro) and in preclinical models,” adds Ellen Roche, PhD, Professor at MIT, who served as co-senior author on the study with Dr. Nguyen. “By bolstering these models with computational simulations, we can characterize and reproduce patient-specific mechanics and flows and assess the potential effects of various types of intervention pre-procedurally.”
Advertisement
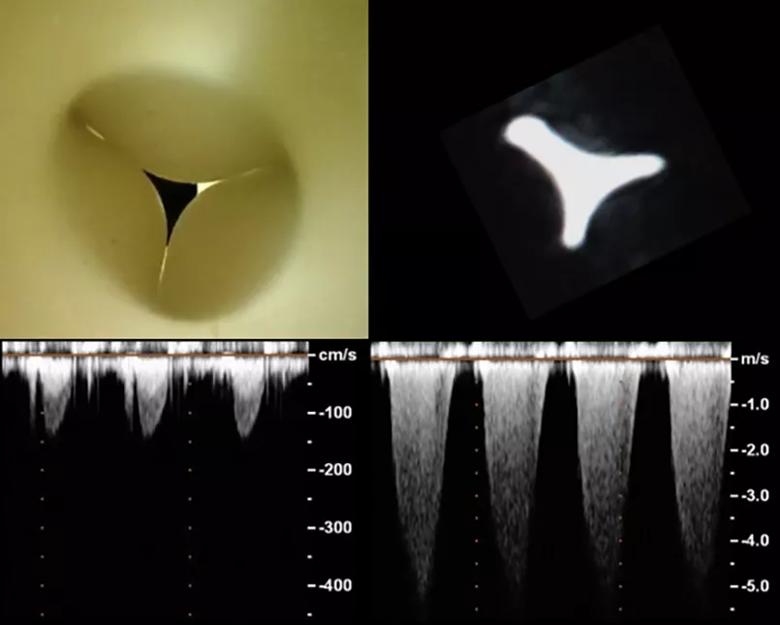
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/2998e413-9d89-4735-8198-61c65e0a0c36/22-HVI-3200902-CQD-Inset_jpg)
Prototype soft robotic sleeve from orthoscopic view (top left) and MRI cross-section (top right). First echo functional measurements (bottom) show that the flow across the stenosis dynamically mimics aortic flow.
Study of the sleeve’s functionality in swine models demonstrated excellent concordance with human aortic stenosis flow, as assessed by 4D blood-flow velocity profiles associated with aortic stenosis, 2D cine MRI imaging, 2D velocity vector mapping, left ventricle catheterization and transepicardial echocardiography.
Orthoscopic video of the robotic sleeve in a pulsatile flow loop.
The team also used CT data to obtain aortic stenosis morphology to design a sleeve with patient-specific parameters. Using only CT data makes the system potentially more applicable to general clinical practice, Dr. Nguyen explains. Comparing outcomes with those using an available commercial device and a nonspecific sleeve (not based on patient parameters) showed that the CT-based sleeve yielded the most accurate aortic stenosis morphology.
Dr. Nguyen foresees multiple applications of the aortic stenosis sleeve model:
Advertisement
Dr. Nguyen adds that his team’s advancement of soft robotics technology can serve as groundwork for others in developing more realistic and dynamic in vivo models of other cardiovascular conditions and other organ systems.
“This body of work reflects an exciting integration of multimodality imaging, computational modeling and biomimetic soft robotics that has resulted in this innovative platform with potential clinical implications,” says Deborah Kwon, MD, Director of Cardiac MRI at Cleveland Clinic, who was not involved in the study. “For example, this platform could augment evaluation of patients with bicuspid aortic valves and severe aortic stenosis who are being considered for transcatheter aortic valve replacement (TAVR). Bicuspid aortic valves are associated with eccentric and complex anatomy, and varying bicuspid valve phenotypes are associated with important imaging features that are unique to each patient. In view of that, this platform could help optimize pre-procedural planning for patients with bicuspid aortic valves in whom TAVR is being considered.”
“The study nicely showcases how multidisciplinary biomedical engineering technologies such as biomechanical device design, advanced medical imaging, 3D printing and computational modeling can synergistically work together,” adds D. Geoffrey Vince, PhD, Chair of the Department of Biomedical Engineering in Cleveland Clinic’s Lerner Research Institute. “I applaud Dr. Nguyen, Dr. Roche and team in pushing the boundaries of designing a patient-specific soft robotic sleeve based on medical imaging data allowing for more realistic hemodynamic profiles in their porcine model of aortic stenosis.”
Advertisement
Advertisement
First full characterization of kidney microbiome unlocks potential to prevent kidney stones
Researchers identify potential path to retaining chemo sensitivity
Large-scale joint study links elevated TMAO blood levels and chronic kidney disease risk over time
Investigators are developing a deep learning model to predict health outcomes in ICUs.
Preclinical work promises large-scale data with minimal bias to inform development of clinical tests
Cleveland Clinic researchers pursue answers on basic science and clinical fronts
Study suggests sex-specific pathways show potential for sex-specific therapeutic approaches
Cleveland Clinic launches Quantum Innovation Catalyzer Program to help start-up companies access advanced research technology