Testing a novel preventive strategy

Myeloid neoplasia arising from inherited bone marrow failure syndromes (IBMFS) in children have poor to fair prognosis even with stem cell transplantation. IBMFS constitute leukemia predisposition syndromes with a high rate of transformation to acute myeloid leukemia (AML). In children with AML, the 5-year survival rate is approximately 65%; however, the prognosis for patients with IBMF-associated AML is poorer due to chemoresistance and the biochemical defects that led to bone marrow failure in the first place. For example, in patients with Shwachman-Diamond syndrome (SDS) complicated by AML, their three year survival is just 11%. The only preventive strategy is stem cell transplant, which carries cost and morbidity and is not necessary for all. Given the poor prognosis, it is imperative to find novel treatment options to prevent AML in this vulnerable population.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
In a new study funded by VeloSano for Kids, researchers plan to apply a nonsense suppressor molecule, Ataluren, as a novel preventive strategy in individuals at high-risk of developing pediatric leukemia.
Patients with severe congenital neutropenia (SCN) have significant reduction in peripheral blood neutrophils and a propensity to infection. In most cases there is a block in neutrophil differentiation at the promyelocyte/myelocyte stage. A rare, genetically transmitted disorder condition, SCN is estimated to affect one in 250,000 live births, but is more common in isolated consanguineous communities.
SCN commonly presents as propensity to infection in infancy. When first described by Rolf Kostmann in the 1950s, most children with SCN did not live past their first birthday. With the use of granulocyte colony stimulating factor (G-CSF) therapy (such as filgrastim or lenograstim) and hematopoietic cell transplantation (HCT) in some cases, the prognosis for patients with SCN has improved. Now as they are surviving longer, malignant transformation of hematopoietic cells impacts survival.
In SCN, secondary nonsense mutations leads to truncation of CSF3R (the receptor for figrastim) in approximately 70% of cases with either myelodysplasia or AML.
Shwachman-Diamond syndrome is also a rare congenital multisystem disorder, with incidence estimated at 1 in 77,000 North American births. The syndrome is characterized by exocrine pancreatic dysfunction, cytopenias and bone abnormalities. Current treatments include G-CSF, stem cell transplantation and pancreatic enzyme replacement therapy.
Advertisement
In SDS, more than 50% of individuals harbor a nonsense mutation in SBDS. Some will acquire somatic TP53 mutations that often precede myeloid neoplasia. Mutant TP53 fails to protect the cell against DNA damage and thus, it promotes the formation of cancer.
“In our study, we will use Ataluren, which has been approved in Europe for the treatment of Duchenne muscular dystrophy, to provide a readthrough of the nonsense mutation to achieve expression of full-length CSF3R and SBDS. We hypothesize that this novel strategy will prevent pediatric leukemia in high-risk individuals,” says principal investigator Hrishikesh Mehta, PhD, Project Staff in Cancer Biology with Cleveland Clinic’s Lerner Research Institute. “We will test the ability of Ataluren to promote full-length SBDS expression in zebrafish, and also determine whether full-length SBDS affects neutropenia and pancreatic atrophy.”
“Zebrafish are an excellent model for the study of inherited neutropenia, as they share a similar genetic structure to humans, as well as many of the same organs and tissues,” says coinvestigator Seth Corey, MD, MPH, Professor in the Department of Pediatric Hematology/Oncology and Cancer Biology at Cleveland Clinic Children’s. Dr. Corey also works with Usua Oyarbide, PhD, and Rebecca Anderson, PhD in the Department of Cancer Biology, who have generated a variety of zebrafish models including ones for Ewing sarcoma and SDS.
Additionally, zebrafish typically lay up to 100 eggs at a time at weekly intervals. Zebrafish embryos develop all main organs by 5 days post-fertilization and are fed externally by this age. “Other models have disadvantages in that they take a long time to breed and develop cancer, and only produce a few offspring at a time, which means we don’t have the numbers needed to power a study. The use of zebrafish can really accelerate the discovery of treatments for inherited cancer predisposition syndromes,” Dr. Corey explains.
Advertisement
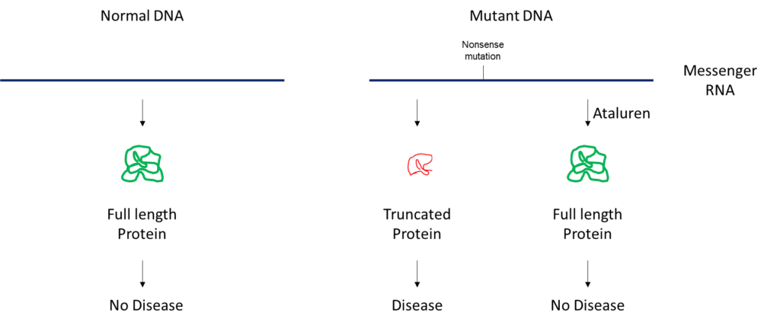
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/180ee96b-a929-473a-8c6c-f9fd98863b9a/image1-1024x427_png)
Please note: This research is funded by a grant from VeloSano Kids. VeloSano Kids was launched in the spring of 2018 as a way for children to get involved with the mission of VeloSano. VeloSano Kids is a year-round, community-driven fundraising initiative, where 100% of the dollars raised supports lifesaving pediatric cancer research at Cleveland Clinic Children’s.
Advertisement
Advertisement

First full characterization of kidney microbiome unlocks potential to prevent kidney stones

Researchers identify potential path to retaining chemo sensitivity

Large-scale joint study links elevated TMAO blood levels and chronic kidney disease risk over time

Investigators are developing a deep learning model to predict health outcomes in ICUs.

Preclinical work promises large-scale data with minimal bias to inform development of clinical tests

Cleveland Clinic researchers pursue answers on basic science and clinical fronts

Study suggests sex-specific pathways show potential for sex-specific therapeutic approaches

Cleveland Clinic launches Quantum Innovation Catalyzer Program to help start-up companies access advanced research technology