Preclinical studies will assess whether method developed for stroke recovery curbs deficits after brain injury
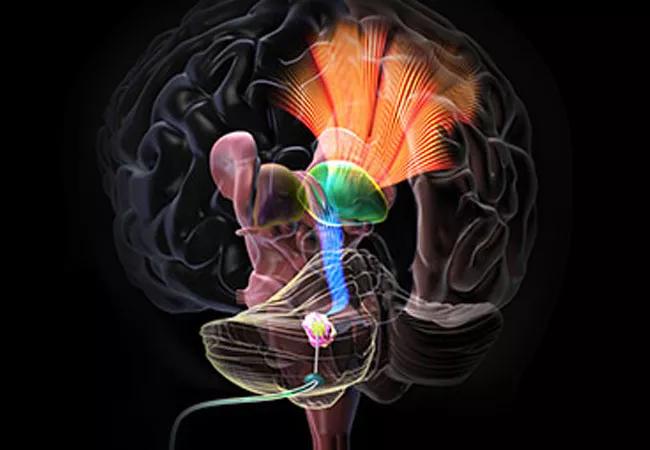
Cleveland Clinic researchers have been awarded a $2.5 million grant from the National Institutes of Health to study the potential therapeutic benefits of deep brain stimulation (DBS) on chronic motor and cognitive deficits associated with post-traumatic brain injury (TBI). The investigations, which were launched July 1, 2020, and will run through June 2025, are being conducted on a laboratory model of TBI and are anticipated to lead to clinical trials.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“This work is in large part an extension of the promising research we have conducted for more than a decade on DBS for stroke rehabilitation,” says co-principal investigator Kenneth Baker, PhD, a researcher in the Department of Neurosciences in Cleveland Clinic’s Lerner Research Institute. “Although there are important differences between TBI and stroke, there are also fundamental common processes involved in chronic recovery, leading us to anticipate that the techniques we’ve developed will benefit TBI rehabilitation.”
The Cleveland Clinic research team is part of a three-institution consortium examining potential therapeutic roles of DBS for TBI. Researchers at the other institutions are focusing on minimally conscious patients and trying to arouse them with stimulation to the central thalamus. In contrast, the Cleveland Clinic investigators are using the cerebellar dentate nucleus as the stimulation focus and concentrating on functional patients with persistent motor and cognitive disabilities.
“No effective therapies currently exist for patients with TBI beyond physical, occupational and speech therapy,” says Andre Machado, MD, PhD, Chairman of Cleveland Clinic’s Neurological Institute and co-principal investigator on the grant. “Even moderately enhancing their function could substantially increase their independence and potential for vocational reentry.”
He adds that the team intends to combine DBS with traditional rehabilitation therapies to prompt an additive or even synergistic effect.
Advertisement
The cerebellar dentate nucleus is an accessible focus of electrical stimulation and a nodal point of the dentatothalamocortical (DTC) pathway, an important tract connecting the cerebellum and the cortex. The pathway has projections to widespread regions in the contralateral brain, including to cognitive, affective and motor areas.
The researchers hypothesize that chronic activation of the DTC pathway with DBS will modulate plasticity of cortical pathways that were spared in brain injury and promote rehabilitation.
“We have good evidence that DBS of the cerebellar dentate nucleus enhances motor recovery in stroke patients, and we have seen indications that cognition is also improved,” says Dr. Baker. “We believe this method promotes functional changes in the brain that translate to improved clinical outcomes.”
The grant-funded work will use a controlled cortical impact rat model of TBI to pursue the following goals:
Advertisement
Because stroke is usually unilateral and TBI is often at least predominantly bilateral, a question that has not been addressed in the stroke investigations is whether unilateral DBS will be sufficient for optimal TBI rehabilitation. The research will compare the effects of unilateral and bilateral DBS.
Another interesting issue is whether DBS over a limited time period will provide enduring benefits. With DBS for Parkinson disease and essential tremor, therapeutic effects stop after stimulation ends. But the team’s experience with DBS and stroke (currently in a phase 1 clinical trial [NCT02835443] with Dr. Machado as principal investigator) has shown that patients tend to reach a plateau in improvement after 5 to 8 months of DBS, after which recovery appears to persist after DBS is turned off.
“We expect that DBS will stimulate the brain to undergo long-lasting reorganization,” Dr. Baker adds. “Our research aims designed to characterize brain changes as well as cellular and molecular changes will help us assess such structural reorganization.”
Advertisement
Advertisement

An argument for clarifying the nomenclature

An expert talks through the benefits, limits and unresolved questions of an evolving technology

Recommendations on identifying and managing neurodevelopmental and related challenges

Phase 2 trials investigate sitagliptin and methimazole as adjuvant therapies

Aim is for use with clinician oversight to make screening safer and more efficient

Rapid innovation is shaping the deep brain stimulation landscape

Study shows short-term behavioral training can yield objective and subjective gains

How we’re efficiently educating patients and care partners about treatment goals, logistics, risks and benefits