Does the clinical benefit manifest electrophysiologically?
By Raghavan Gopalakrishnan, PhD, and Andre Machado, MD, PhD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Post-stroke pain syndrome (PSPS) is an intractable disorder characterized by unrelenting chronic pain and hemiparesis. While traditional analgesia-based therapeutic approaches (e.g., opioids) have largely failed, integrative approaches targeting affective-cognitive spheres have started to show promise.
Recently, a Cleveland Clinic team demonstrated that deep brain stimulation (DBS) of the ventral striatal area, a key node in the networks that modulate emotion, significantly improved the affective sphere of pain and quality of life in patients with PSPS1 (see coverage of that study here). We subsequently examined whether this observed clinical improvement was reflected in the electrophysiological correlates and whether they could serve as objective biomarkers of affective pain response.
To test this hypothesis, we recorded neural substrates of pain anticipation using magnetoencephalography (MEG), a neuroimaging modality that records magnetic fields produced by neural currents with millisecond precision, in 10 patients with PSPS. Recordings were made preoperatively and postoperatively in the “DBS off” and “DBS on” states. Visual countdown cues (numbered 3, 2 and 1 in triangles in Figure 1 below) evoked anticipation as patients awaited a painful stimulus (PS) or nonpainful stimulus (NPS) to their nonaffected or affected extremity (in separate paradigms). Whole-brain event-related responses, a series of fluctuations in neural currents evoked by anticipatory cues, were examined. The responses have specific functional relevance and are labeled P1, N1, P2, N2, etc., depending on the timing and polarity relative to cue onset.
Advertisement
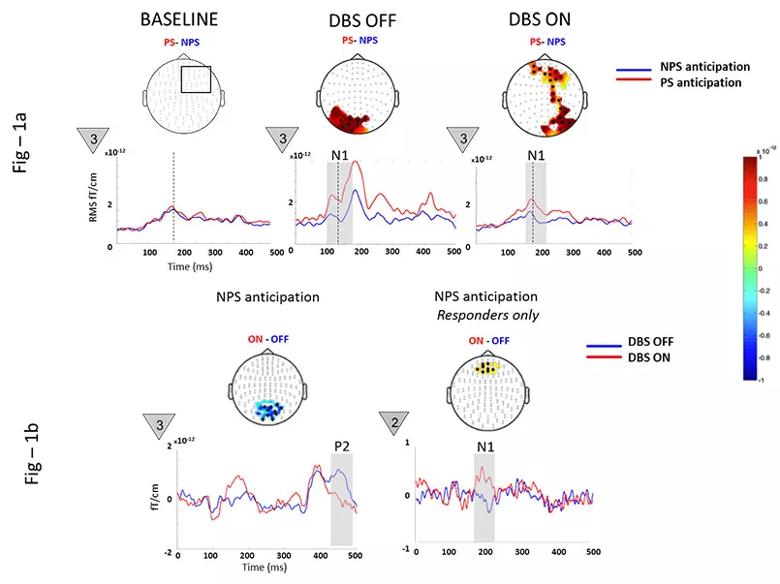
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/2599779e-0562-4958-a915-34b17b79125f/17-NEU-4472-Machado-DBSforPSPS-inset_jpg)
Figure 1. Magnetoencephalographic neural substrate recordings of pain anticipation in 10 patients with post-stroke pain syndrome before and after deep brain stimulation (DBS). See text for details.
Preoperatively, NPS anticipation was remarkably similar to PS anticipation (as shown in the left portion of Figure 1a), possibly due to loss of salience in a network saturated by pain experience. Postoperatively, DBS significantly modulated the early N1 during NPS anticipation, consistent with networks involving restoration of salience and discrimination capacity (Figure 1a, middle and right). Additionally, DBS suppressed the posterior P2 (aberrant anticipatory anxiety) while enhancing the anterior N1 (cognitive and emotional regulation) in treatment responders (Figure 1b).
We conclude that DBS-induced changes in event-related components reflect treatment effects and could serve as biomarkers for treatment efficacy.
Reference
Dr. Machado (machada@ccf.org) is a neurosurgeon and Chairman of Cleveland Clinic’s Neurological Institute. Dr. Gopalakrishnan (gopalar@ccf.org) is Principal Research Engineer in Cleveland Clinic’s Center for Neurological Restoration. They acknowledge Richard Burgess, MD, PhD, of Cleveland Clinic’s Epilepsy Center, for contributions to the work reported here.
Advertisement
Advertisement

An expert talks through the benefits, limits and unresolved questions of an evolving technology

Recommendations on identifying and managing neurodevelopmental and related challenges

Phase 2 trials investigate sitagliptin and methimazole as adjuvant therapies

Aim is for use with clinician oversight to make screening safer and more efficient

Rapid innovation is shaping the deep brain stimulation landscape

Study shows short-term behavioral training can yield objective and subjective gains

How we’re efficiently educating patients and care partners about treatment goals, logistics, risks and benefits

An expert’s take on evolving challenges, treatments and responsibilities through early adulthood