Advertisement
More of the same or truly something new and helpful?
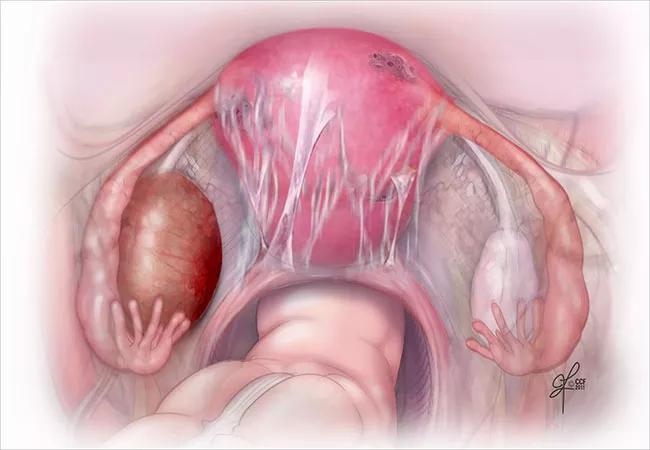
By Rebecca Flyckt, MD, and Mark Dassel, MD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
In July, the FDA announced approval for elagolix (Orlissa™, AbbVie) for the treatment of moderate to severe endometriosis pain, a widely-anticipated release which comes with a flurry of media attention. As the first new endometriosis treatment in over a decade, clinicians are now asking whether elagolix truly represents a novel therapeutic option or is simply more of the same for the millions of women who struggle with this painful disease.
In 2014, a phase II trial funded by AbbVie demonstrated an acceptable efficacy and safety profile for elagolix. The phase III clinical trials of elagolix, involving approximately 950 women ages 18-49, were published in the New England Journal of Medicine in May 2017 (Taylor et al, NEJM 2017). This publication reported on the findings from two six-month, randomized, double-blind, placebo-controlled clinical trials [EM-1 (NCT01620528) and EM-2 (NCT01931670)].
As an example, EM-1 showed that at three months, 46.4 percent of women in the lower-dose group and 75.8 percent in the higher dose group reported reduced dysmenorrhea, compared with 19.6 percent in the placebo group (P < 0.001%). Similar findings were reported in EM-2. For nonmenstrual pelvic pain improvements in EM-1, the three arms reported 50.4 percent vs 54.5 percent vs 36.5 percent (P < 0.001%), respectively. The responses at three months with respect to dysmenorrhea and nonmenstrual pelvic pain were sustained at six months. An extension study which allowed up to 12 months of total treatment was recently published from the same group in Obstetrics and Gynecology in July 2018 (Surrey et al, Obstet Gynecol 2018).
These trials have been some of the first to examine prolonged use of this medication. The phase III trials and extension studies showed improvement in dysmenorrhea as well as nonmenstrual pelvic pain over up to 12 months of use for both the 150mg once daily and 200 mg twice daily formulations of elagolix compared with placebo. For example, in the extension study, 75-78 percent, 67-69 percent and 58-60 percent of women taking the 200mg twice daily dose reported improvement in dysmenorrhea, non-menstrual pelvic pain and dyspareunia, respectively.
Advertisement
The GnRH antagonist formulation known as elagolix has been studied since 2008 as a potential target for endometriosis and uterine fibroids. It competitively binds GnRH receptors in the pituitary gland and results in downregulation of estrogen. Like some other endometriosis treatments such as GnRH agonists, elagolix relies on suppression of ovarian estrogen production via the hypothalamic-pituitary-ovarian axis. Unlike injected GnRH agonists, elagolix is an oral formulation that exerts action that is nearly immediate and has no initial pituitary flare, and also results in estrogen suppression that is dependent on the dosage used. Because it is an oral formulation, elagolix can be more readily discontinued in the case of medication intolerance. Typical use for leuprolide acetate for depot suspension (Lupron Depot®) is six months (longer if add-back norethindrone is used). However, the FDA has approved elagolix for use up to 24 months at the lower (150mg once daily) dose. The 200mg twice daily dose is restricted to six months duration as is any use in women with moderate or severe hepatic impairment.
Elagolix has side effects similar to Lupron Depot, also manufactured by AbbVie. Because both medications work via decreased estrogen production, side effects and adverse effects related to dose-dependent hypoestrogemia are relevant. Hot flushes (24-46 percent of subjects compared to 9 percent of placebo group), night sweats and headaches were reported at both doses of elagolix. The drug also was shown to elevate serum lipids, increase hepatic transaminases and decrease bone density. There were no adverse endometrial effects noted in the trials.
Decreased bone mineral density (BMD) from baseline dual-energy X-ray absorptiometry (DXA) scans was observed in the higher dose of elagolix and was a reason for medication discontinuation in 3.6 percent of subjects in the twice daily arm. These concerns may constrain its long-term utility in a manner similar to current use of GnRH agonists for endometriosis. For example, in study EM-1, compared to placebo, the mean change from baseline in lumbar spine BMD at six months was -0.9 percent (95% CI: -1.3, -0.4) for elagolix 150mg once daily and -3.1 percent (95% CI: -3.6, -2.6) with elagolix 200mg twice daily. The percentage of subjects with greater than 8 percent BMD decrease at any time point in the extension study was 8 percent and 21 percent with once and twice daily dosing, respectively. Although rebound bone density improvements have been established in women after discontinuation of GnRH agonist treatment, it has not yet been proven that losses of bone density in women attributed to elagolix will be fully reversible; future fracture risk is unknown.
The Institute for Clinical and Economic Review (ICER) released a final report in August highlighting perceived limitations in the evidence for long-term efficacy and safety of elagolix. ICER’s independent, evidence appraisal committee voted that the evidence for elagolix was insufficient to demonstrate a net health benefit compared to no treatment. The committee also voted unanimously that the evidence was insufficient for a net health benefit versus either GnRH agonist (leuprolide acetate) or hormonal contraceptives. The ICER report cited concerns with potential hepatic toxicity and unknown long-term effects of the drug. Women on elagolix did conceive during the phase III trial and effects on the fetus are unknown. Patient advocacy groups at the ICER meeting, though appreciative of increased awareness and education surrounding endometriosis as a chronic and debilitating health condition, were vocal regarding concerns that approval of elagolix may prevent women suffering from endometriosis from accessing more effective treatments, subspecialty and multidisciplinary care, and expert surgical management for their disease.
Advertisement
Further, concerns exist regarding the study design of the clinical trials that led to FDA approval of elagolix. In addition to being exclusively funded by the pharmaceutical manufacturer, there were no comparison arms with other standard endometriosis treatments. Therefore no comparison was made to other medication treatments that have been shown effective for the treatment of endometriosis, for example, birth control pills, medroxyprogesterone acetate (Depo-Provera®), progestin-only intrauterine devices, GnRH agonists, or norethindrone acetate. These medications have known side effect and safety profiles and may be lower cost to payers and consumers. Prior small studies comparing these agents with elagolix did not show superiority of the GnRH antagonist.
Further, the clinical trials of elagolix included women who had a diagnosis of endometriosis within the last 10 years, but did not mention endometriosis staging (severity). Even with the extension study, elagolix has only been studied for one year of use, so long-term follow-up is lacking. The long-term safety and efficacy of this drug as well as effects in women who become pregnant unknowingly while on treatment are as yet unknown. The FDA recommends consistent use of other forms of contraception in women taking elagolix.
Treatment with elagolix for one year may cost approximately $10,000, which limits affordability and access for many women. By working with some insurance companies, the manufacturer has taken steps to make the medication more affordable to patients.
Elagolix has shown efficacy for endometriosis-associated pain and expands the available options for women suffering from this disease. As one in 10 women has endometriosis, the application of a treatment such as elagolix may have wide ranging implications.
Taking a balanced perspective on elagolix may best guide clinicians in using this new treatment. It remains to be seen whether the lower dose offers improvements of continuous oral contraceptives and whether the higher dose offers improved long-term patient satisfaction, safety and efficacy in comparison to Lupron Depot.
Therefore, it is prudent to attempt treatment with standard algorithms for hormonal management as a starting point for the majority of women and use elagolix only with biopsy-proven disease that is refractory to other treatments.
It is also important for clinicians to know when women suffering from endometriosis will benefit from consultation with an endometriosis specialist, who may be able to offer combined therapies, including surgical excision, medical therapy, physical therapy and other multidisciplinary pain-directed treatment strategies.
Advertisement
Advertisement

How we create obstacles for sexual, reproductive and menopausal healthcare despite our best intentions

One approved non-hormonal therapy and another on the horizon reduce vasomotor symptoms

Some post-menopausal patients may benefit from treatment

Study shows higher rates of complications, laparotomies among non-white women
Proper diagnosis and treatment require a careful mix of patient and clinical considerations

Study uniquely powered to compare adverse effects
What is female hypoactive sexual desire disorder and how is it treated?

Indications and best-practice recommendations for the use of androgen therapy