Treatment being offered in cases where medical and hormonal management was not successful
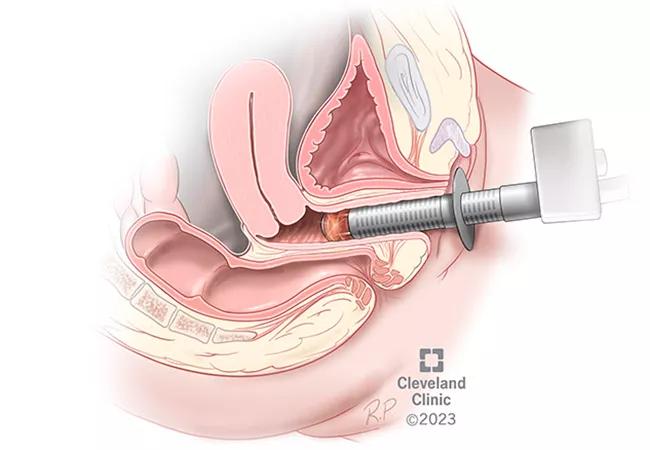
Cleveland Clinic researchers are exploring the long-term safety and efficacy of the MonaLisa Touch — a fractional microblative CO2 laser — for the treatment of genitourinary syndrome of menopause or vaginal atrophy.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The condition is characterized by thinning, drying and inflammation of the vaginal walls, a result of estrogen loss and most often occurs after menopause. For many women, vaginal atrophy causes painful intercourse as well as urinary symptoms, including more frequent urination.
Treatment options for this condition include vaginal moisturizers and water-based lubricants, which can be very effective, according to Shannon Wallace, MD, a specialist in female pelvic medicine and reconstructive surgery at Cleveland Clinic’s Ob/Gyn & Women’s Health Institute.
“Data has also shown that estrogen cream has high efficacy in this patient population,” she says. “However, there are some patients whose conditions are refractory to vaginal estrogen therapy, or who would prefer not to use estrogen because they have a history of breast, ovarian or uterine cancer that is hormone dependent.”
Initial studies suggest that energy-based therapies, such as the MonaLisa Touch, have treatment promise for these patients. Since it is not yet FDA-approved for this use, however, Dr. Wallace does not recommend this approach as a first-line treatment for vaginal atrophy. At Cleveland Clinic, the MonaLisa Touch currently is offered to patients with either refractory vaginal atrophy or vulvar dystrophies who have tried medical and hormonal management without success.
This device, which delivers laser energy to the targeted area, is a short, in-office procedure that requires no downtime. The procedure lasts two to three minutes, and Dr. Wallace says that it is not painful. Patients often describe it as a warm sensation in the vagina.
Advertisement
“Essentially, the laser energy makes small cuts in the vaginal wall that increases collagen and elastin,” explains Dr. Wallace. “It brings cells that create new tissue to the surface of the vagina, so that it can remodel that vaginal tissue. It increases moisture and tissue thickness to reform the vaginal tissue to what you see in premenopausal women or those who have been exposed to estrogen.”
Among eligible patients, the Cleveland Clinic team typically administers three laser treatments every four to six weeks. If improvement is observed over the course of those sessions, patients will then undergo yearly maintenance therapy. However, since this device is a recent innovation, the optimal number of treatment and maintenance sessions has not been established. Dr. Wallace and colleagues are studying this as a part of their research.
The fact that this is a fast, in-office procedure with no recovery time also makes it a very appealing treatment for patients, says Dr. Wallace.
“It is a convenient procedure that doesn’t require daily use of medications or creams, which can be cumbersome and difficult to adhere to for some patients,” she explains. “Therefore, the likelihood of success and adherence with the laser treatment is higher because the time commitment required is much less.”
While this is currently an out-of-pocket expense for patients, Dr. Wallace and colleagues hope to get data that “reflects the efficacy and safety that we’ve seen in our patients, so that insurance companies will cover this for more women.”
Advertisement
Dr. Wallace and her team are developing a database of patients who received the treatment at Cleveland Clinic and are following them over time to gather long-term outcomes and publish their findings in the near future.
“Smaller observational studies suggest that this approach is a safe and potentially effective intervention for genitourinary syndrome of menopause,” Dr. Wallace says. “It is an exciting and new technology that has the potential to be a life-changing option for these patients.”
“And so, we are pioneering this research at Cleveland Clinic to hopefully gain FDA approval for use in this patient population.”
Advertisement
Advertisement

Approximately 500 million people globally are experiencing 'period poverty'

Mode of delivery does not affect patient satisfaction
Education, lifestyle modifications and new drug therapies can help

A case-based discussion of efficacy, eligibility and use

One approved non-hormonal therapy and another on the horizon reduce vasomotor symptoms
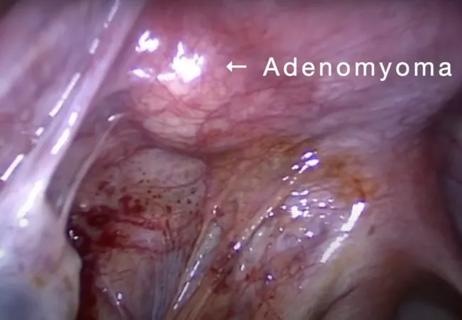
Laparoscopic surgery provides relief for teenage patient with adenomyoma and endometriosis

A large nodule had strangulated the patient’s ureter and invaded the vagina, bladder and rectum
Standardizing obstetrics and gynecology education while promoting patient safety