Optimizing reach to important targets should be a top priority
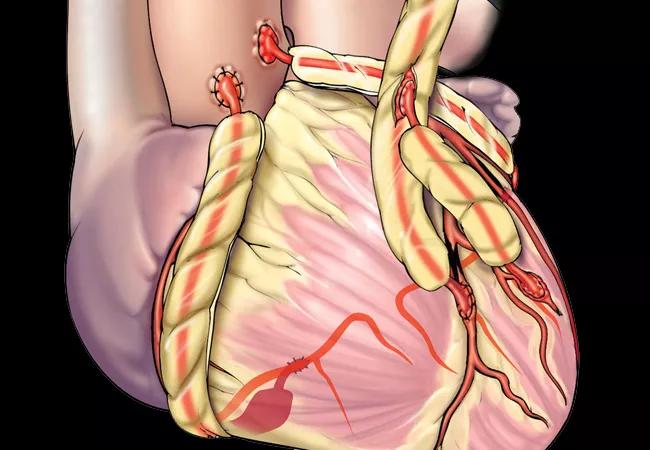
Among coronary artery bypass grafts (CABG) using bilateral internal thoracic arteries (BITAs), right ITA (RITA) grafting achieves high long-term patency independent of its inflow configuration. That’s a key finding from the largest series reported to date of BITA grafts with evaluable angiograms, recently published online in the Annals of Thoracic Surgery.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“When conducting BITA, priority for RITA should be to achieve a configuration that optimizes its reach to important coronary targets without tension and to maximize supply to the myocardium,” says the study’s first author, Cleveland Clinic cardiothoracic surgeon Faisal Bakaeen, MD. “We found that inflow conduit configuration has less of an effect on outcomes than the outflow target.”
Venous grafting has a standard inflow configuration from the ascending aorta, but surgeons use multiple configurations for arterial grafts. That CABG using BITA improves long-term survival over single ITA grafting was established by a large Cleveland Clinic series published more than 20 years ago (J Thorac Cardiovasc Surg 1999;117:855-872). More recently, Cleveland Clinic researchers reported that using BITA to bypass multiple important targets to maximize myocardium perfusion optimizes outcomes (JACC. 2020;75:269-272).
However, little is known about how various RITA inflow configurations affect graft patency. To address this knowledge gap, this study was designed to compare outcomes of various RITA inflow configurations while adjusting for outflow coronary target location.
The study cohort consisted of patients who underwent primary CABG at Cleveland Clinic from 1972 to 2016 and received BITA grafting with available postoperative coronary angiography to assess graft patency (N = 1,331).
Among the LITA grafts, 1,311 (98.5%) were in situ (with 59% of these having an LAD target) and 20 (1.5%) were free graft.
Advertisement
The RITA grafts had more varied configurations. There were 835 in situ inflow grafts (63%), with the LAD as a target in 49% of these. The 496 free grafts (37%) had the following origins:
Overall, RITA patency was 90% at 1 year, 87% at 5 years and 86% at 10 and 15 years. At 15 years, patency rates for various inflow configurations were as follows:
Patency was similar for all these configurations after adjusting for coronary target location and degree of stenosis. However, RITAs grafted to the LAD as the target had fewer occlusions, with patency similar to LITA grafts.
Dr. Bakaeen and his co-authors highlight the following conclusions from this study:
After more than 7,000 cases of BITA grafting at Cleveland Clinic during the study period, the authors recommend the following practices.
Advertisement
“Our findings inform and support flexibility in decision-making in BITA grafting while using best practices,” observes co-author Hiba Ghandour, MD, research fellow in thoracic and cardiovascular surgery at Cleveland Clinic. “They remind us all that the reach of a RITA graft should not determine its target, but the intended target should shape the ITA configuration.”
Advertisement
Advertisement

How Cleveland Clinic is using and testing TMVR systems and approaches

NIH-funded comparative trial will complete enrollment soon

How Cleveland Clinic is helping shape the evolution of M-TEER for secondary and primary MR

Optimal management requires an experienced center

Safety and efficacy are comparable to open repair across 2,600+ cases at Cleveland Clinic

Why and how Cleveland Clinic achieves repair in 99% of patients

Multimodal evaluations reveal more anatomic details to inform treatment

Insights on ex vivo lung perfusion, dual-organ transplant, cardiac comorbidities and more