First-in-world implantation performed at Cleveland Clinic
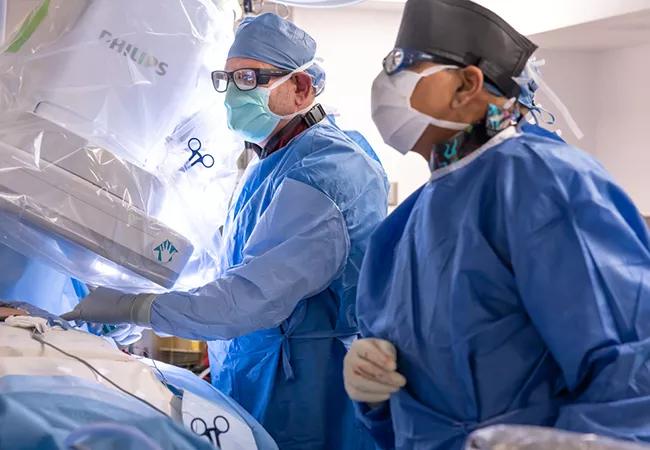
Clinical testing of the first implantable device combining cardiac contractility modulation with cardioversion/defibrillation capabilities is now underway following the world’s first implantation of the device, which took place at Cleveland Clinic.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The investigational OPTIMIZER® Integra CCM-D System (CCM-D System) merges the Optimizer® Smart System, an FDA-approved cardiac contractility modulation (CCM) device, with a new type of implantable cardioverter-defibrillator (ICD). The result is a single device designed to improve the heart’s ability to contract while protecting against sudden cardiac death.
“The device may improve patient heart failure symptoms while at the same time providing therapy for ventricular tachycardia and ventricular fibrillation,” says Niraj Varma, MD, PhD, who performed the first implantation of the device along with his Cleveland Clinic colleague Bruce Wilkoff, MD, in May 2023 (as depicted in the photo above).
Although cardiac resynchronization therapy (CRT) provides defibrillation and also treats heart failure with low ejection fraction, it is used only in patients with left bundle branch block, who make up about 10% to 15% of the heart failure population. This new device potentially applies to another significant portion of heart failure patients.
In distinction to CRT, which reverses ventricular dyssynchrony by pacing the left ventricle, CCM treats the muscle of the failing heart directly by stimulating the ventricular septum for several hours a day. This is nonexcitatory (i.e., it does not pace the heart) but acts to enhance intracellular calcium release and contractility. Diastolic function also improves. Over time, CCM may reverse the adverse remodeling that has occurred during heart failure, thereby strengthening the heart.
Advertisement
“The concept behind the therapy is intriguing,” says Dr. Varma, staff physician in Cleveland Clinic’s Section of Cardiac Electrophysiology and Pacing and consultant electrophysiologist at Cleveland Clinic London. Dr. Varma is the U.S. national principal investigator of the INTEGRA-D study, a multicenter trial evaluating the device that was launched with the first-in-world implantation at Cleveland Clinic.
When viewed as a defibrillator, the CCM-D System is similar in size to other ICDs on the market and is the first to be rechargeable. Whereas current defibrillator batteries need to be replaced every seven to 12 years, the rechargeable defibrillator is expected to last 20 years with only one hour of recharging per week.
“If successful, this will be the first major innovation in defibrillator technology in 20 years,” Dr. Varma notes.
Additionally, the device requires only two leads, a concept that appeals to Dr. Wilkoff, Director of Cardiac Pacing and Tachyarrhythmia Devices at Cleveland Clinic and the institution’s site principal investigator for INTEGRA-D.
“We previously needed to put in two devices — a CCM device and a defibrillator — to achieve better heart function and prevent sudden death,” Dr. Wilkoff says. “Both have leads and need battery changes. This requires multiple procedures that increase the risk of complications, including infection and bleeding. Although antibiotic pouches can reduce infection rates, infections are costly, painful and dangerous. Device extraction, particularly in the setting of an infection, is expensive and potentially hazardous. Anything we can do to reduce the number of procedures and leads reduces the risk of complications, lowers cost and improves care value.”
Advertisement
INTEGRA-D is designed as a prospective, single-arm trial in which the CCM-D System will be implanted in 300 eligible patients at up to 75 centers nationwide.
The goal is to demonstrate whether the CCM-D System can safely and effectively convert induced ventricular fibrillation and spontaneous ventricular tachycardia and/or ventricular fibrillation episodes in patients with stage C or D heart failure who remain symptomatic despite guideline-directed medical therapy. Patients must have a left ventricular ejection fraction of 40% or less and cannot be eligible for CRT. Eligible candidates must be free from prior devices with one exception: a single-chamber defibrillator with a battery nearing the end of its lifespan.
Because the CCM component of the system has already been approved, FDA evaluation is focused on the defibrillator and how the two components operate together. “This is a complicated technology because the two circuits exist separately but overlap,” Dr. Varma explains. “The FDA is interested in how the two systems interact and how the rechargeable aspect is working.”
A subset of study participants will be induced into ventricular fibrillation “on the table” in the procedure room. During the follow-up period, the inappropriate shock rate and device-related complications will be evaluated. The follow-up period is expected to last at least two years.
“If the defibrillator performs according to specifications, it will be approved as a defibrillator,” Dr. Varma notes. “It will then be available for established indications for ICD therapies.”
Advertisement
If the CCM-D System becomes commercially available, Dr. Varma expects additional trials to be conducted on other populations with heart failure who might benefit from the device and had not been studied previously, such as patients who need pacing or CRT.
Drs. Varma and Wilkoff report being paid consultants to Impulse Dynamics, the developer of the CCM-D System and sponsor of the INTEGRA-D trial.
Advertisement
Advertisement

Experts are challenging the one-size-fits-all paradigm

Decongestion, therapy titration and readmission rates comparable to those with traditional hospital care

Panel proposes staging and treatment protocols that support harmonized evaluation and management of heart failure in patients with congenital heart disease

Eminent clinician-researcher brings special expertise in nutritional interventions for heart failure

Recent volume and outcomes data in two major realms of cardiovascular care
Volatile organic compounds have potential in heart failure diagnostics
Basic understanding of condition and treatment is lacking
Indications and issues concerning cardiac resynchronization therapy, conduction system pacing