The role of interventional pulmonology in lung nodules/lesions care
Written by See-Wei Low, MD; Sangita Goel, MD; Marcela Azevedo, MD; Sonali Sethi, MD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
With the U.S. Preventive Services Task Force’s publication in 2021 of revised
recommendations for lung cancer screening came the expectation that many more
patients would be eligible for screening — substantially increasing the number of
indeterminate pulmonary nodules identified annually in the United States using low-dose
computed tomography. Because many peripheral pulmonary lesions (PPLs) require biopsy for a definitive diagnosis, minimally invasive diagnostic techniques with outstanding performance and safety profiles are essential.

Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/0c81c867-6c2b-48dd-b944-6027853adcac/22-PUL-3318470-CQD-Robotic-Broncoscopy-1-inset-300x153_jpg)
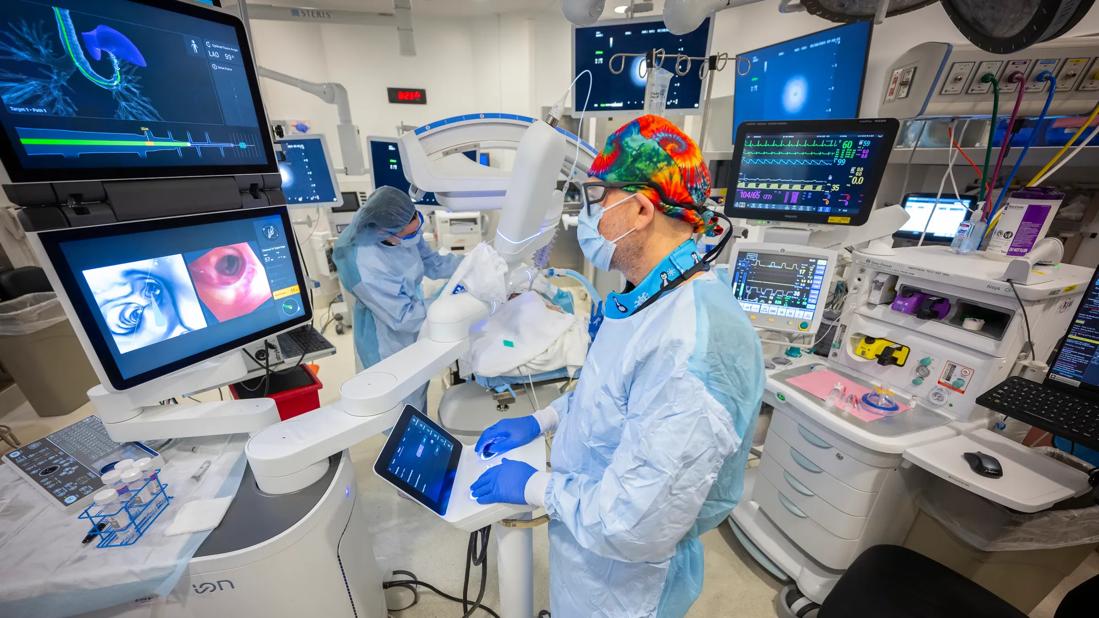
The Monarch Platform from Auris Health Inc. uses electromagnetic navigation guidance. The surgeon uses a video-game-like controller to guide an articulated tube down the patient’s throat. ©Auris Health Inc. Reproduced with permission.
Over the past two decades, several bronchoscopic modalities were introduced — including electromagnetic navigation bronchoscopy (ENB) with digital tomosynthesis correction and an ultrathin bronchoscope with radial endobronchial ultrasound (rEBUS) — to improve the diagnostic yield of PPL biopsies over that derived from biopsies using conventional bronchoscopy. The lower diagnostic yield with conventional bronchoscopy was thought to be due to the inability to directly visualize in the airway periphery, the lack of tip stability in the bronchoscope, catheter articulation and the presence of CT-to-body divergence. The newer modalities increased the diagnostic yield from 54% to 79% and were able to correct CT-to-body divergence using digital tomosynthesis.1 The desire to further improve diagnostic yield resulted in the development of robotic bronchoscopy. In 2018, the FDA approved robot-assisted bronchoscopy (RAB) to facilitate and improve the diagnostic yield of bronchoscopic PPL biopsies.
Advertisement
The RAB systems were designed to reach the peripheral lung airways, similar to the traditional electromagnetic navigation system, but with better stability and steerability during the biopsy. Two RAB platforms are available: the Monarch® Platform (Auris Health Inc., Redwood City, CA) and the Ion Endoluminal System (Intuitive, Sunnyvale, CA); a third platform, Noah Galaxy, is awaiting FDA clearance.
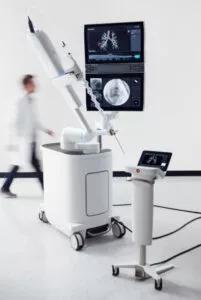
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/e25002a8-4a88-4889-9d27-0b7ef45792a4/22-PUL-3318470-CQD-Robotic-Broncoscopy-2-inset-201x300_jpg)
The Ion Endoluminal System from Intuitive Surgical is a shape-sensing technology that integrates Ion and Siemens Healthineers’ Cios Spin mobile imaging system. The integration offers guidance when preprocedure CT images and intraprocedural imaging indicate differences in nodule locations. © Ion Intuitive. Reproduced with permission.
The Monarch Platform uses electromagnetic navigation guidance, while the Ion Endoluminal System relies on shape-sensing technology. Other subtle differences between the two robots are the scope design (Monarch has a scope-in-scope design, and Ion is a single robotic scope), scope diameter (the outer diameter of the Monarch scope is 4.4 mm and 3.5 mm for the Ion Scope), and the need with Ion to remove the vision probe to insert the confirmation or biopsy tool. More recently, Ion also offers the ability to correct CT-to-body divergence when Ion is integrated with the Siemens Healthineers Cios Spin 3D mobile imaging system.
Many have asked which robots are better suited for reaching peripheral nodules with higher accuracy and whether robots are worth the excitement. Sonali Sethi, MD, responded this way: “Early diagnosis and treatment of lung cancer can dramatically improve outcomes. RAB is evolving into an approach that allows us to offer patients a safe, streamlined and timely diagnosis. There is a lot of enthusiasm, and the robotic future is bright as the field continues to evolve in future directions, including advanced confirmatory imaging and bronchoscopic ablation.”
Advertisement
Multiple studies have reported improved diagnostic yield with these RAB platforms; however, no one has conducted a randomized trial or a comparative study of these two robotic platforms or compared the RAB to the conventional EMN system. Most recently, RAB was demonstrated to be as accurate as CT-guided transthoracic biopsy (overall diagnostic yield was 87.6% for RAB and 88.4% for CTTB) by Lee-Mateus et al. at Mayo Clinic.2
In an abstract recently presented by Low et al. at the World Congress for Bronchology and Interventional Pulmonology (WCBIP) 2022, the Ion platform was demonstrated to have a diagnostic yield similar to that of the ILLUMISITETM platform (Medtronic, Minneapolis, MN) (77% for RAB and 80% for ILLUMISITETM, p = 0.4).3 Comparing diagnostic yield from one study to another is challenging for several reasons, the most important being the lack of a standardized definition of “diagnostic yield.” Understanding these limitations, multiple studies have reported similar diagnostic yields (Monarch Platform, 69.1% to 93%4-6; Ion system, 79.3% to 95%7-9), outcomes and safety of the robots. A head-to-head randomized trial between the robots, but a comparative study is possible.
At Cleveland Clinic, we are privileged to have both RAB platforms. We can integrate real-time imaging during the procedure with Ion utilizing the Cios Spin, as well as incorporate C-Arm Based Tomography (LungVisionTM by Body Vision Medical Inc., Campbell, CA) with the Monarch. We look forward to further improving patient care with an increased diagnostic yield of PPLs, minimizing potential complications and providing ablative therapies bronchoscopically. The ultimate vision of incorporating RAB and real-time imaging into the practice is having the ability to diagnose, stage and treat the PPLs during one procedure while the patient is under anesthesia, leading to efficient and effective care.
Advertisement
References
Advertisement
Advertisement

Caregivers are provided with real-time bronchoscopy patient findings

A review of treatment options for patients who may not qualify for surgery

Milestone minimally invasive surgeries reduce pain and recovery time

Collaborative patient care, advanced imaging techniques support safer immunotherapy management

Our latest performance data in these areas plus HOCM and pericarditis

Rising rates in young miners illustrate the need for consistent prevention messaging from employers and clinicians

Multidisciplinary focus on an often underdiagnosed and ineffectively treated pulmonary disease

Management and diagnostic insights from an infectious disease specialist and a pulmonary specialist