
New insights on effectiveness in patients previously treated with other anti-VEGF drugs

A multitude of subspecialities offer versatility, variety

Evidence mounts that these diabetes and obesity drugs may protect eyes, not endanger them

It’s the first step toward reliable screening with your smartphone
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy

CFH gene triggers the eye disease in white patients but not Black patients
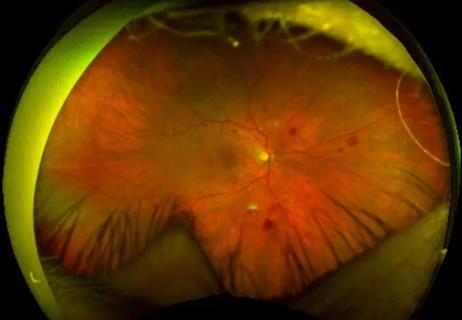
Study identifies factors that may predict vision outcomes in diabetic macular edema

From medication to laser treatment to surgery

Fixational eye movement is similar in left and right eyes of people with normal vision

Watch for sudden unilateral vision loss without pain

Preclinical study shows why it’s critical to consider sex in eye disease research
Advertisement
Advertisement