Novel approach allows activation mapping, VT ablation despite cardiogenic shock
Ablation for ventricular tachycardia (VT) is limited by the need to repeatedly induce VT to map the faulty circuits, potentially causing hypoperfusion that can lead to end-organ damage. Cleveland Clinic has been pioneering the use of the Impella 5.0 temporary left ventricular assist device to provide support in patients who present in cardiogenic shock in need of salvage VT ablation.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Thus far, about 20 patients have undergone successful VT ablation in this setting at Cleveland Clinic, with encouraging outcomes.
“Regardless of Impella support, salvage VT ablation is very challenging, involving high morbidity and mortality,” says Oussama Wazni, MD, Section Head of Cardiac Electrophysiology and Pacing at Cleveland Clinic, who served as lead cardiologist in many of these cases to date, including the one profiled below. “These patients are on the verge of death when they come in, so it is gratifying to find a way to pull them through, eliminate the source of ventricular tachycardia and offer them a better life.”
A recent representative case is profiled below.
A 72-year-old man presented to Cleveland Clinic for recurrent implantable cardioverter-defibrillator (ICD) shocks. He had had a myocardial infarction more than 30 years earlier, followed by coronary artery bypass graft surgery seven years later. He was left with ischemic cardiomyopathy with an ejection fraction of 25%.
One month prior to this presentation, he was admitted to Cleveland Clinic for ICD shocks for VT over the course of five days; the VT was deemed not amenable to ablation due to multiple fast cycle lengths on interrogation. At that time, he was restarted on amiodarone and sent home with an atrial lead to support bradycardia.
During the current presentation, his baseline ECG showed evidence of an old inferior infarct. Echocardiography revealed ejection fraction of 24%, left ventricular diastolic diameter of 6.1 cm, and 2+ mitral regurgitation. Akinesis was observed on the anterolateral, inferior and posterior walls. PET scan showed no ischemia but revealed a greater than 20% scar in the area of the left circumflex artery.
Advertisement
He was admitted overnight for ICD shock, and the heart failure team was consulted. On day 4 of hospitalization, an Impella 5.0 pump was inserted via the right axillary artery by cardiothoracic surgery. This transvalvular flow pump can provide up to 5.0 L/min blood flow for support of left ventricular function. On the following day, the patient was taken to the electrophysiology lab, where VT ablation was successfully performed for sustained hemodynamically unstable VT (see photo below). He was without pulsatile flow for 40 minutes but was supported with 4.6 L/min of continuous flow from the Impella pump. The first ablation terminated the VT, and more extensive ablation was then performed.
Following surgical removal of the Impella device a few days later, the patient recovered and is doing well several months after the procedure.
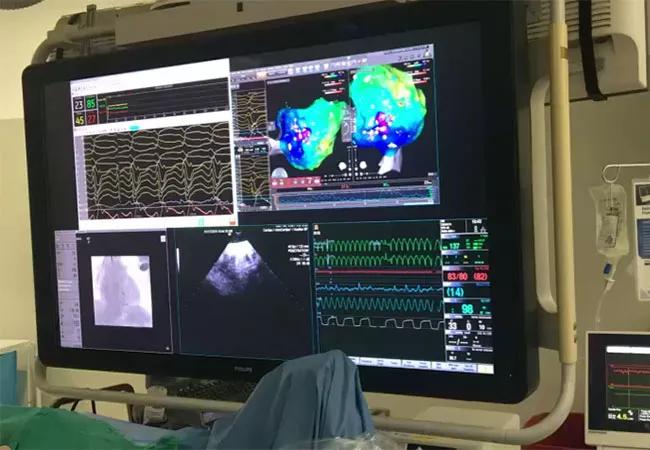
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/d7a2ded1-ab29-4e5b-b19a-811e10864d88/19-HRT-3760-VT-ablation_jpg)
Photo from the electrophysiology lab during the patient’s procedure.
Impella support for salvage VT ablation requires several important elements, as outlined below.
1) Adequate Impella support. The Impella device generally is implanted two to four days before VT ablation and remains in place for a few days afterward to ensure support in case of complications. Complications in our experience have included vasoplegia and hematoma related to subclavian artery access.
“In such cases, one must be prepared to escalate to a durable left ventricular assist device or transplantation if needed,” says cardiothoracic surgeon Edward Soltesz, MD, MPH, Surgical Director of Cleveland Clinic’s Kaufman Center for Heart Failure Treatment and Recovery and lead surgeon in the case above.
Advertisement
2) General hemodynamic monitoring. Although the Impella device allows maintenance of patients without VT, they are still in cardiogenic shock, so close hemodynamic monitoring is required at all times. Risk is compounded by the need to induce VT to perform activation mapping.
3) Neuromonitoring. Cerebral oximetry is used to determine oxygen saturation in the brain and ensure adequate perfusion during the procedure. When VT is induced, cerebral oximetry may be as low as 50% saturation or reflect a 20% drop from baseline. We have maintained patients without pulsatile flow for up to one hour, and all have emerged cognitively intact.
4) A multidisciplinary team. This is essential to manage these patients. At Cleveland Clinic, a cardiothoracic surgeon, a heart failure specialist and a cardiothoracic anesthesiologist are present with the electrophysiologist who performs the ablation, and cardiothoracic surgery and heart failure management teams are closely involved throughout the patient stay.
According to Dr. Wazni, Cleveland Clinic’s ongoing goals for use of this strategy include identifying better hemodynamic and inflammatory markers to better define risk and prognosis.
Advertisement
Advertisement
Large retrospective analysis may prompt prospective studies
How to talk about lifetime risk, treatment goals, Lp(a) testing, statin skepticism and more
A scannable recap of recent volumes and clinical metrics from Cleveland Clinic
Cleveland Clinic reports first U.S. series focused on use in this challenging setting
Large series confirms early and long-term survival advantages over partial pericardial resection
AVANT GUARD trial extends first-line role for ablation beyond paroxysmal atrial fibrillation
Maintain a high index of clinical suspicion and consider the underlying etiology
Protocol adoption at Cleveland Clinic sharply raised share of transferred patients getting timely PCI