Answer lies in a single mutation in a key protein
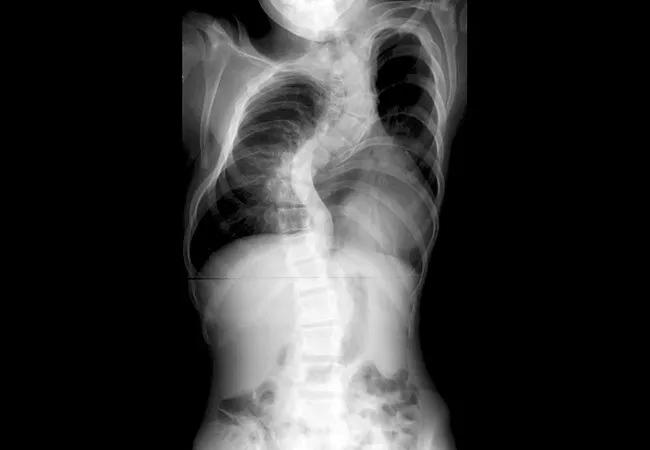
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/1caf89fb-b026-4bda-a260-7894a6789c58/17-CCC-447_jpg)
17-CCC-447
Basic research performed in the laboratory of molecular cardiologist Jun Qin, PhD, has uncovered the basis for how a single mutation in the protein filamin results in the devastating genetic disease known as frontometaphyseal dysplasia (FMD).
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
FMD and other filamin-related disorders are X chromosome-linked diseases affecting 1 in 100,000 births. Males with FMD usually die in utero: Those who survive die early with complications in multiple organs. Girls born with FMD suffer from milder birth defects.
The protein filamin helps maintain the shape of cells by binding to many other proteins, forming a network. Abnormal filamin present in FMD and other filamin- inherited disorders leads to defects in the skeleton, heart, nervous system, digestive system and eyes.
Using highly sensitive biophysical studies, Dr. Qin and research associate Sujay Ithychanda, PhD, discovered that a single mutation in one of filamin’s 2,647 amino acids turns the protein from rigid to floppy. This causes a threefold weakening in its ability to attach to protein partners in the cell, resulting in the deformation of the structure of bone and tissue throughout the body.
“Usually when a protein becomes floppy, it does not function at all. In this case, it binds poorly with other proteins,” Dr. Ithychanda explains.
Their finding, published in the Journal of Biological Chemistry, is the first to explain the basic molecular pathogenesis for FMD.
For years, Drs. Qin and Ithychanda have studied how filamin makes physical connections to other proteins using nuclear magnetic resonance imaging and other leading-edge tools to examine its building blocks at the molecular level. Their work has resulted in the identification of seven docking locations in filamin for other proteins. “The role of these protein-protein connections in human diseases has been obscure until now,” says Dr. Ithychanda.
Advertisement
Filamin is an ideal protein for such research because it is involved in many diseases, including cancer, diabetes and heart disease. “Focusing on this single protein enables us to study multiple organ systems,” says Dr. Qin. “Our recent finding will provide a valuable framework as we start to look at how other mutations in the protein affect underlying biochemical processes and disease progression.”
Curing FMD would require the use of CRISPR technology to alter the defective gene in utero before the disease occurs. However, the fundamental understanding of how a mutation in filamin results in this disease, as illustrated in this study, paves the way for development of drugs to improve the quality of life for patients with this, and other, devastating disorders.
Advertisement
Advertisement
First full characterization of kidney microbiome unlocks potential to prevent kidney stones
Researchers identify potential path to retaining chemo sensitivity
Large-scale joint study links elevated TMAO blood levels and chronic kidney disease risk over time
Investigators are developing a deep learning model to predict health outcomes in ICUs.
Preclinical work promises large-scale data with minimal bias to inform development of clinical tests
Cleveland Clinic researchers pursue answers on basic science and clinical fronts
Study suggests sex-specific pathways show potential for sex-specific therapeutic approaches
Cleveland Clinic launches Quantum Innovation Catalyzer Program to help start-up companies access advanced research technology