
Treatments can be effective, but timely diagnosis is key

While logistical questions remain about RPM, its benefits for both patients and caregivers are abundantly clear

A mindset shift has changed the way pulmonologists both treat and define PFF
Emerging trends are transforming the classification, diagnosis, therapies and management of ILD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
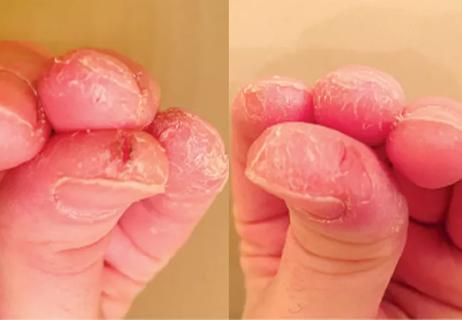
Look beyond skin changes
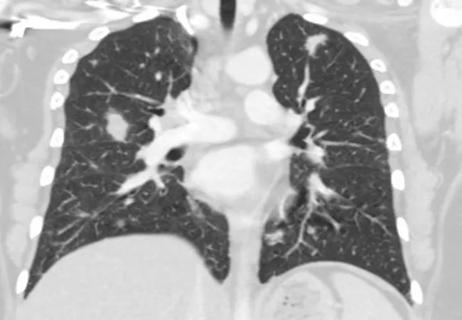
Multidisciplinary treatment of rare and complex immunodeficiencies
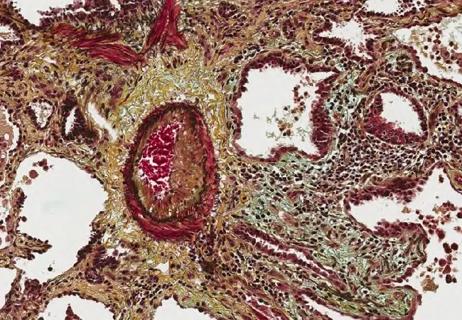
May allow greater independence and quality of life

Multidisciplinary approach can uncover unsuspected diagnoses
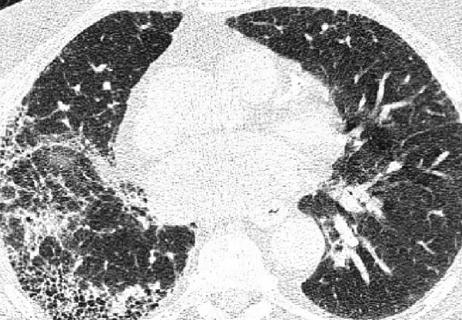
New pathways to consider in the treatment of SSc-ILD
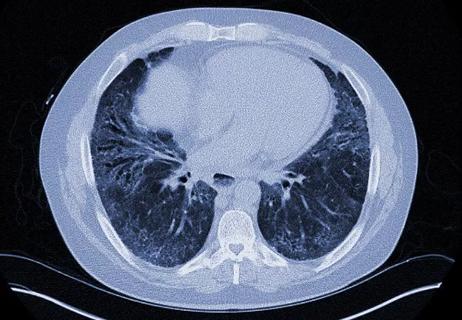
Nintedanib significantly reduces loss of lung function
Advertisement
Advertisement