Management of high-risk RMSK in the pre-and current eras of neoadjuvant therapy
Tyrosine kinase inhibitors (TKI) in the neoadjuvant setting are associated with greater use of partial nephrectomy (PN) when treating patients diagnosed with renal masses in a solitary kidney (RMSK), according to the findings of a new Cleveland Clinic study. While randomized, controlled trials are needed, these findings support the use of TKI, still considered an experimental neoadjuvant therapy, in select patients with RMSK.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
They published their findings in European Urology Oncology.
This is the latest RMSK series from Steven C. Campbell, MD, PhD, Worapat Attawettayanon, BS, and colleagues, who recently updated their experience with this challenging patient population in 2022. The research team published a comprehensive review that assessed outcomes from more than 1,000 patients diagnosed with RMSK at Cleveland Clinic between 1975 and 2022. The 2022 study is among the largest series of RMSK outcomes to date and the first to include all RMSK patients — not just those who underwent successful PN.
It also inspired new questions about the management of patients with the most complex cases of RMSK.
“Coming out of that study, we asked ourselves, ‘What can we learn about the most challenging RMSK cases?’ These are the cases with the highest risk of postoperative complications or loss of renal function. The ones with a RENAL score of 10 to 12,” Dr. Campbell explains.
These are the cases where there is no room for error. Complications could mean converting to radical nephrectomy and, ultimately, progression to dialysis. Though precarious, high-complexity RMSK is relatively uncommon. Still, Dr. Campbell says, most urologists will likely encounter a few of these cases in their professional lifetime.
Cleveland Clinic began administering TKI as a neoadjuvant therapy for RMSK in 2009. The medication is an angiogenesis inhibitor, blocking the vascular endothelial growth factor receptors involved in the growth of clear cell renal cell carcinoma — a tumor subtype that accounts for about 70% of kidney cancers.
Advertisement
In the right patients, it offers a clear benefit. “You might have a 7 cm tumor shrink to a 5 cm tumor, which sounds modest, but this is significant if it offers greater accessibility, making the surgery less technically challenging, and allowing us to save more renal parenchyma,” he explains.
The research team extracted data from the Cleveland Clinic kidney cancer database (1999-2022), resulting in 499 records with an available RENAL score, a validated metric to classify renal tumors based on quantifiable characteristics, and focused on patients with scores of 10 to 12, resulting in 133 patients for analysis.
They then divided the dataset into two cohorts: patients who predated the availability of TKI (1999-2008) (N = 80) and patients managed when neoadjuvant TKI could be considered, based on the date of their surgery (2009-2022) (N = 53).
Their analysis found that 43% (23/53) of patients with clear cell histology in the TKI era were selected for the neoadjuvant therapy. Overall, these 23 patients had a greater median tumor diameter and RENAL score than their counterparts not selected for the neoadjuvant therapy. These were the most challenging of cases. And, generally, they responded well to TKI therapy. A decrease was noted in the median tumor diameter (from 7.1 cm to 5.6 cm) and RENAL score (from 11 to 9). Tumor volume was reduced by 59%.
Nearly all of these patients (21/23), or 91%, were able to be managed with PN. Overall, in the TKI era, 48/53 (90%) of patients were managed with PN. In contrast, in the pre-TKI era, only 65% (52/80) were able to be managed with PN.
Advertisement
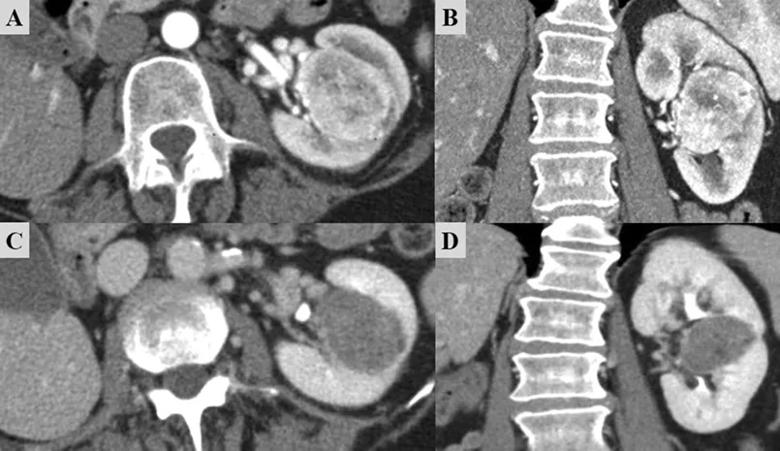
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/6126d576-c9ac-4a75-87ec-18fc45961368/23-URL-4036562-CQD-Inset_jpg)
Figure. Imaging before and after TKI therapy for a patient with biopsy-proven clear-cell RCC in a solitary kidney. (A,B) Before TKI treatment, the tumor was 4.5 cm, entirely endophytic, and adjacent to the main vascular branches coming out of the hilum, with a RENAL score of 11. The patient was then treated with 8 weeks of axitinib. (C,D) Post-TKI imaging shows a smaller tumor size and substantial tumor necrosis; the RENAL score had decreased to 10. The tumor also pulled away from the hilum to some degree. The patient underwent open clamped partial nephrectomy with hypothermia. Pathology demonstrated grade 2 clear-cell RCC with negative margins. Surgery preserved 77% of the glomerular filtration rate, and the patient is cancer-free with stable renal function after 2 yr of follow-up. RCC = renal cell carcinoma; TKI = tyrosine kinase inhibitor.
Image and caption have been republished under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0). Originally published by Attawettayanon W, Yasuda Y, Zhang JH, Kazama A, Rathi N, Munoz-Lopez C, Lewis K, Shah S, Li J, Emrich Accioly JP, Campbell RA, Shah S, Wood A, Kaouk J, Haber GP, Eltemamy M, Krishnamurthi V, Abouassaly R, Weight C, Derweesh I, Campbell SC. Selective Use of Neoadjuvant Targeted Therapy Is Associated with Greater Achievement of Partial Nephrectomy for High-complexity Renal Masses in a Solitary Kidney. Eur Urol Open Sci. 2023 Jun 10;54:1-9.
Dr. Campbell acknowledges that a randomized, controlled trial would offer a higher level of evidence about the use of TKI therapy in this patient population. But their data provide new information on a historically limited dataset, setting the stage for continued investigations.
Advertisement
Still, he advises prudent use of TKI therapy and careful consideration of the cost-benefit for patients. Clear cell histology alone is not an indication for TKI therapy, and if there is limited benefit to downsizing the tumor from the surgeon’s perspective, the patient may proceed with surgery and PN without TKI therapy. But in cases with more complicated tumors, the advantages of TKI cannot be overlooked.
“In cases where the tumor shrinks substantially — well, that can turn an incredibly difficult case into an easy one,” Dr. Campbell concludes.
Featured image: Imaging shows before and after TKI therapy for a patient with biopsy-proven clear-cell RCC in a solitary kidney. See figure for full description.
Advertisement
Advertisement

One pediatric urologist’s quest to improve the status quo

First single-port renal vein transposition reduces recovery time and improves outcomes
Fully-automated process uses preop CT, baseline GFR to estimate post-nephrectomy renal function
Belzutifan superior to everolimus in phase 3 clinical trial
NIH-funded study explores novel MRI technique to stage cystic kidney disease
AI-generated model bests predictive abilities of human experts
Study highlights benefits of nephrologist-led urine sediment analysis
Using sequencing data to identify novel factors linked to kidney disease with unknown origin