Husni lab recently funded by NIH-NIAMS
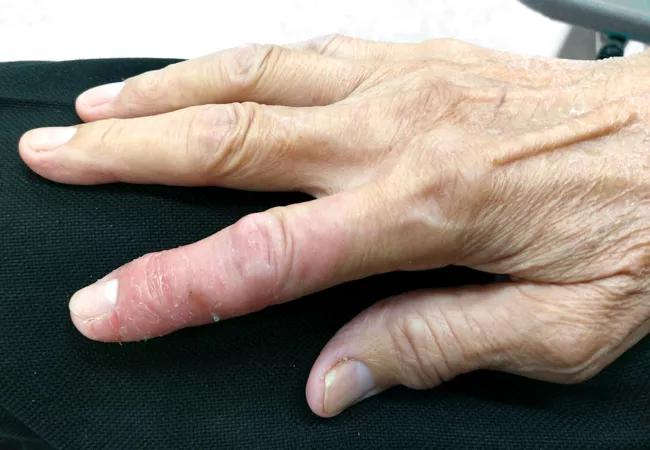
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/1acd1dd0-b8bf-4bc2-9da2-df3179e2886b/17-RHE-1348-Husni-Hero-Image-650x450pxl-1_jpg)
17-RHE-1348-Husni-Hero-Image-650x450pxl
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The Husni Lab is dedicated to advancing discoveries in psoriatic diseases and related comorbidities. Our lab recently received a grant that allows us to focus on a more targeted approach to treatment that may help reduce the signs and symptoms of psoriasis and psoriatic arthritis with fewer side effects. I and co-principal investigator Unnikrishnan M Chandrasekharan, PhD, lead scientist in our lab and staff in the Department of Cellular and Molecular Medicine at Cleveland Clinic’s Lerner Research Institute, are working to define novel disease-specific signaling pathways to allow more precisely targeted anti-tumor necrosis factor (TNF) therapy in psoriatic diseases. Although highly effective in treating psoriatic diseases, the chronic use of anti-TNF therapy can cause unwanted side effects because it also blocks TNF-α, a cytokine important in regulating infection and cancer surveillance.
To that end, our lab is focusing on the cell signaling of TNF-α, as its dysregulation plays a significant role in the skin and joint pathology of psoriatic diseases. Current anti-TNF medications act by binding TNF-α in circulation before it binds to its two immune cell surface receptors, TNFR1 and TNFR2, thus preventing the activity of both receptors. By contrast, our work involves selective inhibition of one of the two TNF-α receptors. This approach will still reduce the signs and symptoms of psoriatic diseases while preserving some protective mechanisms of TNF-α.
Our preclinical models using mice genetically engineered to selectively eliminate TNFR1 or TNFR2 receptors responded differently to a chemically induced psoriasis murine model. The findings suggest that specific blockade of TNFR2 may significantly reduce inflammation and ameliorate signs of psoriatic diseases, while also maintaining normal immune response in the host, including the ability to combat infection and cancer.
Advertisement
Furthermore, we know that some patients do not respond to anti-TNF therapies, and our work has shown that this lack of response may be related to a genetic variant of TNFR2, TNFR2-M196R. Our studies will uncover the mechanisms of TNFR2-M196R that link to this reduced response to anti-TNF agents in certain patients.
The newly funded grant allows us to further develop these findings and ultimately test the hypothesis that inactivation of TNFR2 or chemical inhibition of its signaling intermediates will relieve psoriatic inflammation in patients. Our study also tests whether TNFR2-M196R polymorphism does, in fact, reduce a patient’s response to anti-TNF agents. This may help to predict inadequate responders to anti-TNF therapies early on, allowing more personalized approach in treating psoriatic diseases.
The National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health awarded our lab a $1.8 million R01 grant for five years to investigate the mechanisms of TNFR2 activation and its impact on psoriatic pathogenesis. We plan to continue our translational work in both murine models and our patient biorepository. Selective targeting of TNFR2 is a novel approach and can be applied to improving the safety and precision of treating a broad range of immune-mediated diseases.
Dr. Husni is Director of the Arthritis and Musculoskeletal Center in the Department of Rheumatic and Immunologic Diseases and Cleveland Clinic’s Psoriatic Disease Biobank.
Advertisement
Advertisement
First full characterization of kidney microbiome unlocks potential to prevent kidney stones
Researchers identify potential path to retaining chemo sensitivity
Large-scale joint study links elevated TMAO blood levels and chronic kidney disease risk over time
Investigators are developing a deep learning model to predict health outcomes in ICUs.
Preclinical work promises large-scale data with minimal bias to inform development of clinical tests
Cleveland Clinic researchers pursue answers on basic science and clinical fronts
Study suggests sex-specific pathways show potential for sex-specific therapeutic approaches
Cleveland Clinic launches Quantum Innovation Catalyzer Program to help start-up companies access advanced research technology