Several factors masked the cause of the patient’s chronic condition
Editor’s Note: This article was first published in the Cleveland Clinic Journal of Medicine.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
By Soumya Chatterjee, MD, MS, FRCP, FACP, FACR
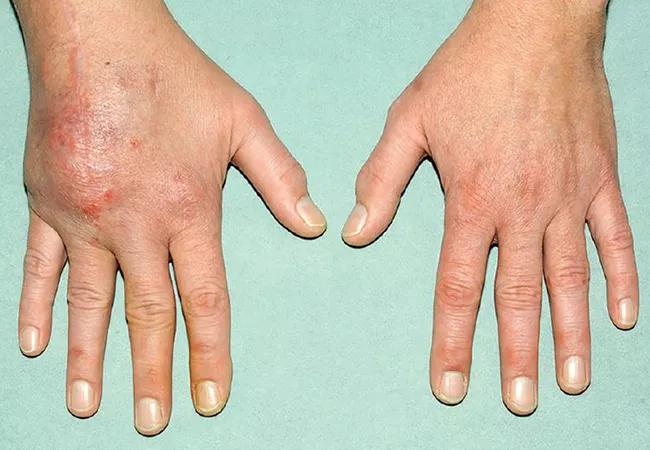
A 48-year-old male industrial electrician presented for consultation with a five-year history of pain and swelling of the dorsum of the right hand, with limited mobility of the right wrist and flexion of the fourth finger. The hand inflammation had begun as a red nodule on the dorsum of the second metacarpophalangeal joint, which slowly progressed to a soft, doughy-looking, subcutaneous mass encompassing the dorsum of the right hand.
The patient’s previous mycobacterial and fungal cultures, stains for organisms and purified protein-derivative testing for tuberculosis were negative. Chest radiography and thoracic computed tomography were normal. The pathologist suggested the diagnosis of chronic cutaneous sarcoidosis with tenosynovitis.1 The patient was treated with prednisone (15 mg daily) and methotrexate (15 mg weekly) with daily folate supplementation. However, the hand pain and swelling persisted, and new areas of inflammation appeared. Biopsies from the right olecranon bursitis two years later (patient still on prednisone and methotrexate) and from a right forearm mass three years later again revealed noncaseating granulomata with negative cultures.
When we saw the patient, because previous mycobacterial and fungal cultures were negative on three occasions, and because histologic re-evaluation of previous slides revealed noncaseating granulomatous inflammation with multinucleated giant cells, the initial diagnosis of sarcoidosis was still considered likely. Hence, infliximab, a monoclonal antibody that binds to tumor necrosis factor-alpha (TNF-alpha), was added to treat the presumably refractory sarcoidosis. 2-4
Advertisement
One week after the second infusion, the swelling and redness of the right hand became noticeably worse. Infliximab was discontinued, and diagnostic tenosynovectomy was performed, along with another deep biopsy of the right dorsal hand mass (the patient’s fourth biopsy). Rice bodies were extruded through the surgical incision. Stains for bacteria, mycobacteria and fungus were negative. However, an acid-fast bacillus grew on culture and was identified as Mycobacterium marinum by sequence identification. Infliximab was presumed to have unmasked the underlying chronic infection.5, 6
Methotrexate was discontinued, and the patient was weaned from prednisone. The microbiology laboratory was alerted to include sustained cultures at lower temperatures, and M marinum grew at 23 days. Based on in vitro minimum inhibitory concentration values, the patient was started on rifampin, ethambutol and azithromycin.
Eight months later, the patient required surgical repair of a ruptured right middle finger extensor tendon. One year after that repair, while still receiving antibiotics, he experienced swelling of the right olecranon bursa, requiring excision. Ziehl-Neelsen stain of smears from mycobacterial growth indicator bottles revealed M marinum microcolonies, and the tissue tested positive for M marinum by sequence identification. At that time, the patient was started on trimethoprim-sulfa methoxazole based on its reported efficacy in eradicating treatment-refractory M marinum infection, and the other antimycobacterial antibiotics were discontinued. Treatment was continued for two years after the complete resolution of symptoms and findings.
Advertisement
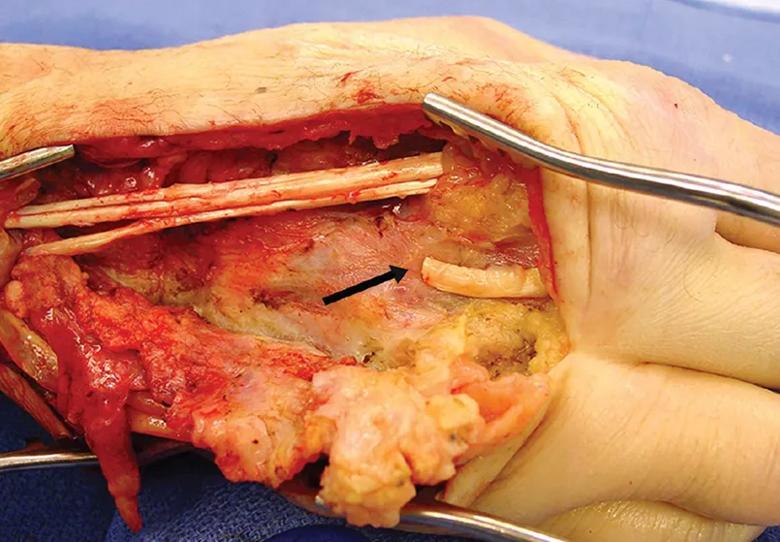
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/519a9bc5-cc5a-4bfc-b93b-43f29ecf266e/23-RHE-3524512-Inset_jpg)
The patient required surgical repair of a ruptured extensor tendon (arrow) of the right middle finger.
M marinum is a nontuberculous mycobacterium that usually infects humans by exposure to contaminated fresh or marine water through damaged skin. Notably, apart from this patient’s exposure as an industrial electrician to cooling water pools for induction furnaces, he had no clear history of exposure to any recognized water source for M marinum infection.
M marinum infection can present as a nodular granulomatous dermatitis resembling cutaneous sarcoidosis7 or as tenosynovitis. It often starts as a solitary violaceous or red plaque or nodule, sometimes with a crusted or verrucous surface, which can spread along lymphatics, similarly to lymphangitic sporotrichosis.7
In immunocompetent patients, the infection is usually limited to the skin and soft tissues, whereas disseminated M marinum infections can occur in patients with HIV/AIDS and other immunocompromised states. Infected synovial fluid, bursae or tendon sheaths may contain rice bodies (small, loose pellets that resemble polished grains of white rice). They are composed of cellular debris or sloughed infarcted synovium, surrounded by fibrin, sometimes with a collagenous core. In the past, rice bodies were mainly associated with poorly controlled rheumatoid arthritis. Currently, they are more often associated with chronic (especially mycobacterial) infection.
M marinum grows optimally at 32°C (89.6°F), and growth is inhibited at 37°C (98.6°F). The infection involves the colder parts of the body, especially the extremities. Ziehl-Neelsen stain of biopsy specimens is rarely positive. Therefore, cultures should be obtained and observed for at least six weeks and are ultimately positive in up to 80% of patients.7 In our patient’s case, all previous cultures before presentation to our clinic were reportedly negative, presumably because of the low number of organisms in the clinical specimens and incubation of the tissue at 37°C (not at 28°C–32°C) in the microbiology laboratory due to lack of suspicion for M marinum infection. TNF-alpha plays an essential role in granuloma formation, limiting the spread of a mycobacterial infection. When a TNF-alpha inhibitor was started in this patient, it permitted rapid multiplication of organisms, leading to a positive acid-fast bacillus culture and detection of M marinum by sequence identification, ultimately revealing the diagnosis.5,6
Advertisement
M marinum should be considered when a patient presents with nonhealing nodular skin lesions in the distal extremities. The differential diagnosis of such lesions should also include granulomatous fungal infections such as sporotrichosis, blastomycosis, coccidioidomycosis, cryptococcosis and histoplasmosis. Other chronic skin infections with a similar presentation are cutaneous tuberculosis, other nontuberculous mycobacterial infections, leishmaniasis and nocardiosis. Cutaneous sarcoidosis should be considered in the differential diagnosis, as in our patient’s case, but sarcoidosis remains a diagnosis of exclusion.
In our patient, the absence of exposure to fish tanks or outdoor water and the repeatedly negative mycobacterial and fungal cultures deterred us from initially suspecting the correct diagnosis. Eventually, an unexpected response to TNF-alpha antagonist therapy facilitated recognition of the correct diagnosis. Infections with M marinum in patients receiving TNF-alpha antagonists and other biologic agents have been well described.5,6
Therapy of M marinum is not standardized.7 The organism is generally resistant to isoniazid, pyrazinamide and para-aminosalicylic acid. In a study of 63 clinical cases, rifamycins and clarithromycin were the most potent agents against M marinum.8 There is no consensus on the duration of therapy, as there are no randomized controlled trials. Combination therapy is recommended for immunosuppressed patients. In addition, surgical debridement is often needed to facilitate cure. In immunocompetent patients, treatment should continue for at least two months after all skin lesions have healed. Immunocompromised patients require therapy with two agents for at least six months after the resolution of all skin lesions.
Advertisement
1. Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med 2007; 357(21):2153–2165. doi:10.1056/NEJMra071714.
2. Noiles K, Beleznay K, Crawford RI, Au S. Sarcoidosis can present with necrotizing granulomas histologically: two cases of ulcerated sarcoidosis and review of the literature. J Cutan Med Surg 2013; 17(6):377–383. doi:10.2310/7750.2013.13035.
3. Adler BL, Wang CJ, Bui TL, Schilperoort HM, Armstrong AW. Anti-tumor necrosis factor agents in sarcoidosis: a systematic review of efficacy and safety. Semin Arthritis Rheum 2019; 48(6):1093–1104. doi:10.1016/j.semarthrit.2018.10.005.
4. Baughman RP, Judson MA, Lower EE, et al. Infliximab for chronic cutaneous sarcoidosis: a subset analysis from a double-blind randomized clinical trial. Sarcoidosis Vasc Diffuse Lung Dis 2016; 32(4):289–295. pmid:26847095.
5. Berman HS, Thomas L, Beynet D, Gates G, Shi V, Hsiao J. Fish tank granuloma during tumor necrosis factor-alpha inhibitor treatment: a case report and literature review. Dermatol Ther 2020; 33(6):e14058. doi:10.1111/dth.14058.
6. Winthrop KL, Yamashita S, Beekmann SE, Polgreen PM; Infectious Diseases Society of America Emerging Infections Network. Mycobacterial and other serious infections in patients receiving anti-tu-mor necrosis factor and other newly approved biologic therapies: case finding through the Emerging Infections Network. Clin Infect Dis 2008; 46(11):1738–1740. doi:10.1086/587989.
7. Akram SM, Aboobacker S. Mycobacterium marinum. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2021.
8. Aubry A, Chosidow O, Caumes E, Robert J, Cambau E. Sixty-three cases of mycobacterium marinum infection: clinical features, treatment, and antibiotic susceptibility of causative isolates. Arch Intern Med 2002; 162(15):1746–1752. doi:10.1001/archinte.162.15.1746.
Advertisement

A conversation with Leonard Calabrese, DO

The case for continued vigilance, counseling and antivirals

High fevers, diffuse rashes pointed to an unexpected diagnosis

No-cost learning and CME credit are part of this webcast series

Summit broadens understanding of new therapies and disease management

Program empowers users with PsA to take charge of their mental well being

Nitric oxide plays a key role in vascular physiology

CAR T-cell therapy may offer reason for optimism that those with SLE can experience improvement in quality of life.