Proposing a classification system for irAEs
The emergence of immune-related adverse events (irAEs) from cancer immunotherapy represents a formidable challenge for rheumatologists. A new classification system for irAEs may help to focus discussions of these heterogeneous complications.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Currently, ICI therapy may be used to treat over 40% of all tumors. An estimated 10%-20% of cancer patients treated with immune checkpoint inhibitor (ICI) therapy may develop a serious immune-related adverse event (irAE). As the use of ICIs expands, irAEs may become more commonplace.
“Since 2011, the field of cancer immunotherapy has grown rapidly, with many new drugs and strategies being approved for many different malignancies. Unfortunately, unleashing the immune system allows it to not only battle malignancies, it also allows autoimmune diseases to flourish,” says Leonard H. Calabrese, DO (@LCalabreseDO). “The field of irAEs related to ICI therapy is growing quickly, and, as rheumatologists, we need to be prepared to manage it.”
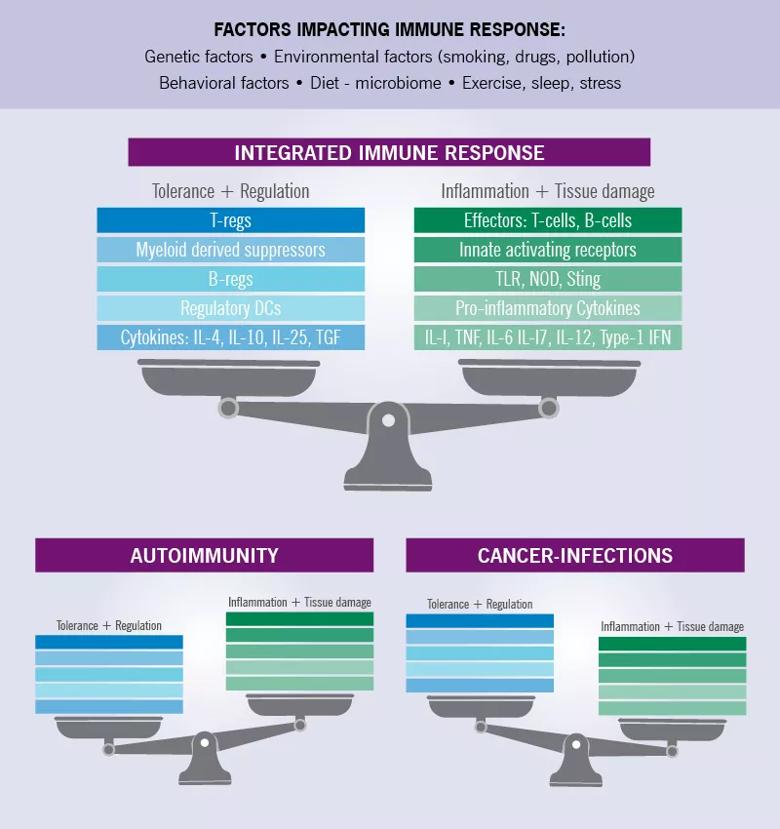
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/33b1d2ed-1f36-45f0-8296-076b282b6ea9/ImmuneRel-advrs-CancerImlmunothrpy_jpg)
The integrated immune response in its homoeostatic state is a balance of complex tolerogenic and inflammatory forces, each contributing to optimal surveillance and patrolling functions designed to detect and dispatch danger while preserving the integrity of the host. The end product of the response is influenced by a variety of factors including the hosts genetic background and external variables and events that potentially can alter the immune system. In cancer and chronic infections there is an imbalance of depressed local-regional or systemic effector functions allowing continued growth of the tumor or persistence of infection. In autoimmunity, inflammatory effector function is overactive relative to tolerance and regulation. Both immunotherapy with ICIs and immunosuppressive therapies can shift this balance, with ICIs increasing local/systemic inflammatory reactions and immunosuppressive therapies such as glucocorticoids and cDMARDS and bDMARDS, and other therapies suppressing inflammation with the theoretical possibility of compromising antitumoral effects.
Advertisement
In a recent editorial, published in the Annals of the Rheumatic Diseases, Dr. Calabrese offers three categories of immune-related adverse events (irAEs).
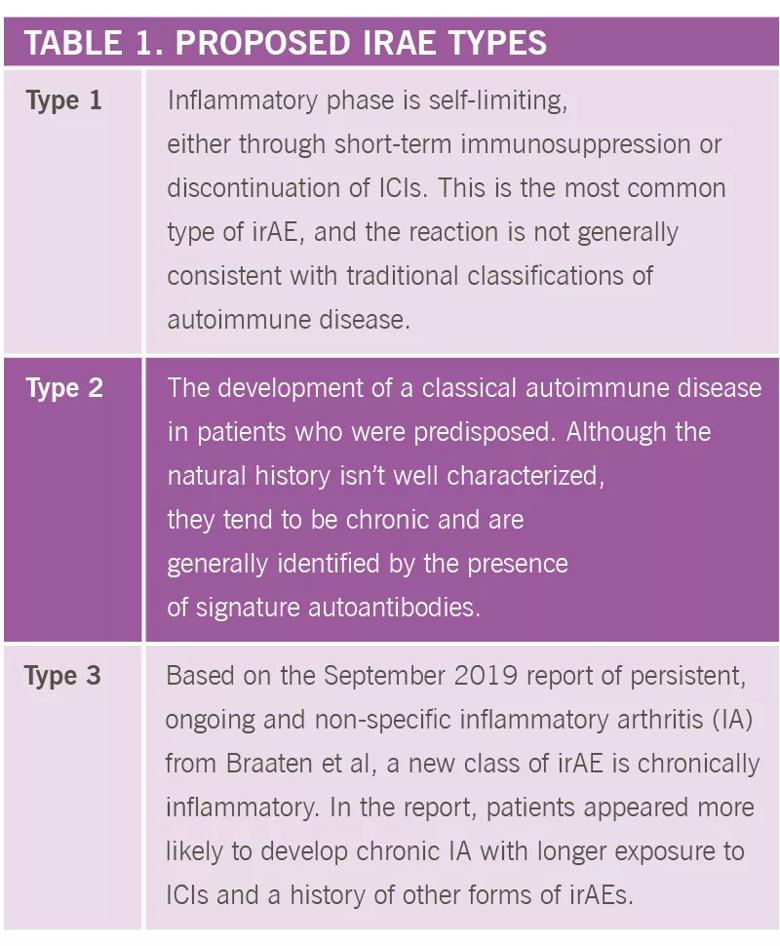
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/93935b91-43da-4c74-adca-d21cf7f78c2d/19-RHE-1355-ImmuneRel-advrs-CancerImlmunothrpy-table_jpg)
“When we confront irAEs in the clinic, the primary objective is to facilitate optimal cancer treatment with immune-based therapies whenever possible,” Dr. Calabrese says. “We aim to balance the anti-inflammatory and immunosuppressive effects of the chosen irAE therapy while preserving the anti-tumor effects of the immunotherapy.”
It appears that some irAEs (type 3) may become chronic, and possibly permanent, inflammatory diseases. The first line of therapy is steroids, with the objective being to employ the smallest effective dose. Concerns have been raised about the possibility that steroids may reduce the effectiveness of ICIs against cancer; however, there is no convincing data to prove that low doses of steroids are detrimental, Dr. Calabrese notes. In the Braaten et al study, disease modifying anti-rheumatic drugs (DMARDs) were used in 40% of patients with ICI-induced IA, including biologic DMARDs in 11% of patients, without impacting cancer outcomes.
“The overall treatment approach must be based on a balance between the benefits and risks, and may continue to evolve as we learn more about ICI-related irAEs,” Dr. Calabrese notes. “Further studies are urgently needed.”
Advertisement
Advertisement

The case for continued vigilance, counseling and antivirals

High fevers, diffuse rashes pointed to an unexpected diagnosis

No-cost learning and CME credit are part of this webcast series

Summit broadens understanding of new therapies and disease management

Program empowers users with PsA to take charge of their mental well being

Nitric oxide plays a key role in vascular physiology

CAR T-cell therapy may offer reason for optimism that those with SLE can experience improvement in quality of life.

Unraveling the TNFA receptor 2/dendritic cell axis