Treatment strategies require understanding of pathomechanisms
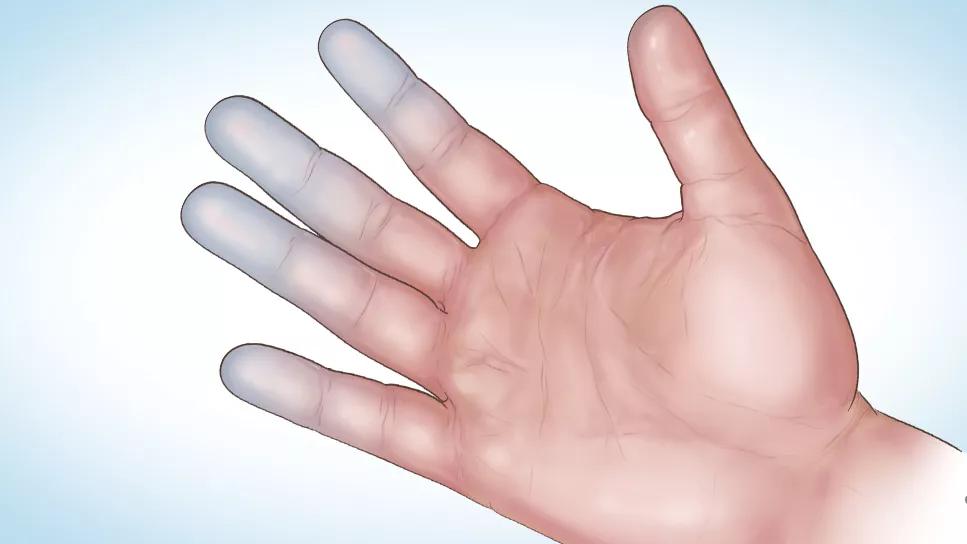
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/969bd003-09dc-47a4-8365-586b702a8593/24-RHE-4630253-CQD-967x544-1_jpg)
Medical illustration of hand with discolored fingers
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Erythromelalgia (EM) is a rare neurovascular disorder characterized by spontaneous episodes of redness, heat, swelling and intense burning pain in the extremities. Symptoms are exacerbated by warmth, pressure, exercise, stress, alcohol, caffeine or dependency of the affected extremity, whereas cooling and limb elevation relieve the pain. EM can be primary or secondary.
Primary EM is caused by mutations in the voltage-gated sodium channel a-subunit gene SCN9A [1], which may be familial (autosomal dominant inheritance) or sporadic. Secondary EM can result from myeloproliferative disorders, small fiber neuropathy, medications (calcium channel blockers, bromocriptine, pergolide and topical isopropanol), mushroom or mercury poisoning, and various autoimmune diseases.
Raynaud’s phenomenon (RP) is characterized by constriction of small arterioles, triggered by cold exposure or emotional stress, causing pallor of the fingers or toes, followed by cyanosis and/or rubor. RP may also be primary (Raynaud’s disease) or secondary. The latter is associated with systemic sclerosis, mixed connective tissue disease (MCTD) or lupus, and less often with thromboangiitis obliterans, thoracic outlet syndrome, paraneoplastic syndrome, hypothyroidism or use of vibrating machinery or vasoconstrictors.
A case illustrating the coexistence of EM and RP and its possible pathomechanisms is described here.
A 49-year-old female with mixed connective tissue disease (MCTD) presented to the rheumatology clinic with episodic attacks of RP and EM. Her illness started when she was 45 with a new onset of RP and inflammatory polyarthritis. She had dilated capillary loops and cuticular hemorrhages on nailfold capillaroscopy, a positive antinuclear antibody (1:320, homogeneous pattern), and anti-U1-RNP antibody (3.1 AI; Ref range: 0–0.9 AI).
Advertisement
She also suffers from severe attacks of EM. When trying to keep her feet and toes warm, if her socks are too thick or too tight, her feet become red, hot, swollen, painful, and throbbing. Both her legs and feet become intermittently red and hot, but never simultaneously. Warm water and compression gloves cause hot, swollen, painful and red hands. Spicy food causes her face, neck, chest and arms to turn red, splotchy and painful. Extreme temperatures make outdoor activities difficult. Sleep is disrupted as her body is cold, but her feet are “on fire.”
Cold exposure and stress also trigger RP, but calcium channel blockers cause severe facial flushing from worsening EM [2–4].
As she had no myeloproliferative disorder, aspirin was ineffective for her EM. Laboratory tests ruled out other causes of EM.
EM and RP have specific initiating events and clinical manifestations, although the patient’s primary and secondary forms rarely coexist [4–8]. Both start with vasospasm, followed by reactive hyperemia, which is more pronounced in EM. These two seemingly opposite conditions may share some common causative and pathophysiologic mechanisms [9].
In a subset of EM, increased cutaneous vascular tone lowers the morning basal temperature [9], followed by reactive hyperaemia later in the day, as in the rubor phase of RP. The initial vasoconstriction results from structural or functional alterations in the endothelium and/or smooth muscle or cutaneous microvascular innervation [9]. Cutaneous vasoconstriction causes the ischemic cells to switch to anaerobic respiration, accumulating nociceptive inflammatory mediators that open up arteriovenous anastomoses [10].
Advertisement
This scenario may explain why some EM cases respond to calcium channel blockers, similar to RP.
Conversely, the vasoconstriction model does not address perpetually vasodilated EM patients [5], where calcium channel blockers exacerbate symptoms [2–5]. Vascular studies in warm and cool environments could distinguish the two subtypes of EM and determine whether vasodilation or vasoconstriction will control symptoms [5].
Optimal management strategies for patients concurrently affected by EM and RP require a more in-depth understanding of the pathomechanisms underlying these two paradoxically opposite conditions.
[1] Yang Y, Wang Y, Li S, et al. Mutations in SCN9A, encoding a sodium channel alpha subunit, in patients with primary erythermalgia. J Med Genet 2004;41:171–4.
[2] Brodmerkel Jr GJ. Nifedipine and erythromelalgia. Ann Intern Med 1983;99:415.
[3] Fisher JR, Padnick MB, Olstein S. Nifedipine and erythromelalgia. Ann Intern Med 1983;98:671–2.
[4] Sunahara JF, Gora-Harper ML, Nash KS. Possible erythromelalgia-like syndrome associated with nifedipine in a patient with Raynaud’s phenomenon. Ann Pharmacother 1996;30:484–6.
[5] Berlin AL, Pehr K. Coexistence of erythromelalgia and Raynaud’s phenomenon. J Am Acad Dermatol 2004;50:456–60.
[6] Kalgaard OM, Seem E, Kvernebo K. Erythromelalgia: a clinical study of 87 cases. J Intern Med 1997;242:191–7.
[7] Lazareth I, Priollet P. Coexistence of Raynaud’s syndrome and erythromelalgia. Lancet 1990;335:1286.
[8] Slutsker GE. Coexistence of Raynaud’s syndrome and erythromelalgia. Lancet 1990;335:853.
Advertisement
[9] Littleford RC, Khan F, Belch JJ. Skin perfusion in patients with erythromelalgia. Eur J Clin Invest 1999;29:588–93.
[10] Belch J. Temperature-associated vascular disorders: Raynaud’s phenomenon and erythromelalgia. In: Tooke JELG, editor. A Textbook of Vascular Medicine. London: Arnold; 1996. p. 329–52.
This article was originally published in Joint Bone Spine, Volume 90, Issue 4, 2023, 105561. https://doi.org/10.1016/j.jbspin.2023.105561.
Advertisement
Advertisement
Treatment insights from the ninth recorded EMPD case in 50 years
Scleromyxedema may cause extracutaneous symptoms
First-in-human phase 1 trial induced loss of function in gene that codes for ANGPTL3
Unraveling the TNFA receptor 2/dendritic cell axis
Nasal bridge inflammation, ear swelling and neck stiffness narrow the differential diagnosis
Genetic testing at Cleveland Clinic provided patient with an updated diagnosis
Major study demonstrates importance of having a multidisciplinary approach to treatment for large, locally advanced tumors
Researchers Assess Real-Life Experiences of Patients Treated Outside of Clinical Trials