Longer-term confirmation of VALOR-HCM could yield a noninvasive therapy for severe obstructive HCM
Sixteen weeks of treatment with the investigational agent mavacamten significantly reduced the need for invasive septal reduction therapy in patients with intractable symptoms of obstructive hypertrophic cardiomyopathy (HCM) in the multicenter VALOR-HCM trial.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Results of the phase 3 study were reported by principal investigator Milind Desai, MD, MBA, in a late-breaking clinical trials presentation at the American College of Cardiology’s Scientific Session on April 2.
“Adding mavacamten to maximally tolerated medical therapy significantly reduced patients’ eligibility for and/or desire to proceed with septal reduction therapy,” says Dr. Desai, Director of the Hypertrophic Cardiomyopathy Center at Cleveland Clinic. “No approved medical therapies have been developed specifically for hypertrophic cardiomyopathy or evaluated in randomized controlled trials for this condition, so mavacamten shows promise to address an unmet need for a noninvasive treatment in this setting.”
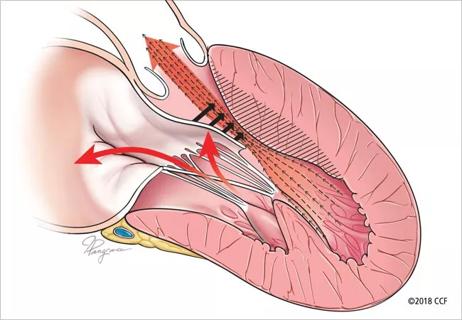
Medical therapy with beta-blockers, calcium channel blockers and/or disopyramide may improve symptoms in patients with moderate left ventricular outflow tract (LVOT) obstruction. These therapies are typically inadequate, however, when patients progress to severe symptomatic obstructive HCM. In such cases, septal reduction therapy (SRT) — either surgical myectomy or alcohol septal ablation — is recommended and has been shown to improve long-term survival and quality of life.
While SRT is highly effective, it is invasive. “Additionally, in order to achieve guideline-recommended optimal results of < 1% operative mortality, it is recommended to be performed at high-volume experienced centers,” Dr. Desai notes. “This potentially reduces its availability to patients.”
Mavacamten is a targeted inhibitor of cardiac myosin designed to reduce the excessive contractility characteristic of HCM. It has previously been shown to improve LVOT gradient, quality of life and physical function in patients with obstructive HCM, “but we wanted to determine its potential to serve as an alternative to septal reduction therapy in patients with severely symptomatic and refractory obstructive HCM,” Dr. Desai explains. “Additionally, it can serve as a complementary therapy to treat these patients and could also have a larger role in patients in whom SRT is not an option.”
Advertisement
VALOR-CHM used a multicenter, randomized, double-blind, placebo-controlled design. The study’s 112 enrollees had documented HCM with a maximum septal wall thickness (determined by core echo lab) of at least 15 mm, or at least 13 mm in those with a family history of HCM. Other key inclusion criteria were:
Patients could choose to proceed to SRT at any time after randomization.
Enrollees were randomized 1:1 to mavacamten 5 mg or matching placebo once daily by mouth. Randomization was stratified by type of SRT recommended (surgical myectomy or alcohol ablation) and by NYHA class.
Echocardiography was performed by the core lab at baseline and every four weeks thereafter through the 16-week treatment period. Measures of LVEF and LVOT gradient at rest and with provocation were used to titrate drug dosage at weeks 4, 8 and 12. Dosage could be reduced to 2.5 mg (starting at week 4) or increased up to 10 mg (starting at week 8) or to 15 mg (starting at week 12). After 16 weeks, all patients had the chance to continue in a long-term extension study.
Advertisement
The primary endpoint was a composite of the patient’s decision to proceed with SRT by week 16 or continued eligibility for SRT (according to 2011 ACC/AHA guidelines) at week 16.
The study was coordinated by the Cleveland Clinic Coordinating Center for Clinical Research (C5Research) and Medpace (Cincinnati, Ohio).
The mavacamten and placebo groups were highly comparable in demographic and clinical characteristics, including age (mean of 60 years), sex (51% male), NYHA class (93% class III or IV) and background medical therapy.
In the study’s intention-to-treat analysis (N = 112), the primary endpoint of proceeding with SRT or continued guideline eligibility for SRT was reached by 43 of 56 patients in the placebo group (76.8%) compared with 10 of 56 patients in the mavacamten group (17.9%) (P < 0.0001).
Of the 10 mavacamten recipients meeting the primary endpoint, two proceeded to SRT by week 16 and eight were guideline-eligible at week 16. Among the 43 placebo recipients meeting the primary endpoint, two proceeded to SRT, 39 were guideline-eligible at week 16, and two had non-evaluable SRT status.
Mavacamten showed significant treatment benefits relative to placebo on each of the study’s secondary endpoints (P < 0.0001 for all):
Notably, 63% of patients on mavacamten improved by at least 1 NYHA class at 16 weeks, and 27% improved by at least 2 NYHA classes.
Advertisement
Adverse event rates were generally comparable between the two treatment arms, with no new safety signals observed and no cases of syncope, heart failure, sudden cardiac death or treatment discontinuation due to LVEF ≤ 30%.
“Patients with severely symptomatic obstructive HCM were significantly less likely to proceed with, or meet eligibility criteria for, septal reduction therapy if they were randomized to 16 weeks of mavacamten rather than placebo,” says Dr. Desai. “This suggests that adding mavacamten to maximally tolerated medical therapy may reduce, delay or serve as an alternative to septal reduction therapy in these patients, although additional data are needed to assess the durability of this effect and the drug’s safety over longer follow-up.”
If the results are confirmed over longer periods, they have important implications for patient care, according to VALOR-HCM coinvestigator Nicholas Smedira, MD, MBA, Surgical Director of Cleveland Clinic’s Hypertrophic Cardiomyopathy Center. “Septal reduction therapy is best performed at experienced centers with mortality rates lower than the national average,” says Dr. Smedira. “However, there are few high-volume centers, and this limits patient access to optimal outcomes. An effective noninvasive alternative would give us a much-needed additional option to offer highly symptomatic patients for whom conventional medical therapy has failed. It would help expand our toolbox of offerings to such patients, in addition to SRT.”
The investigators note several new contributions this study makes to the growing evidence base for mavacamten:
Advertisement
The study was funded by MyoKardia (a wholly owned subsidiary of Bristol Myers Squibb), which is developing mavacamten. Dr. Desai reports that he is a consultant for Bristol Myers Squibb.
Advertisement

5 biggest changes according to Cleveland Clinic experts

AHA scientific statement outlines models for high-intensity staffing, specialized expertise and more

5% of flagged ECGs in real-world study were from patients with previously undiagnosed HCM

Transvalvular LVAD support transforms emergency surgery into urgent procedure

High composite score in myectomy specimens signals worse prognosis
Few patients report left ventricular dysfunction or heart failure after one year
Avoidance of septal reduction therapy continues while LVEF dysfunction remains infrequent

Aortic valve replacement is best option for lowering mortality in this high-risk population