Will support preclinical work in parallel with ongoing human trial
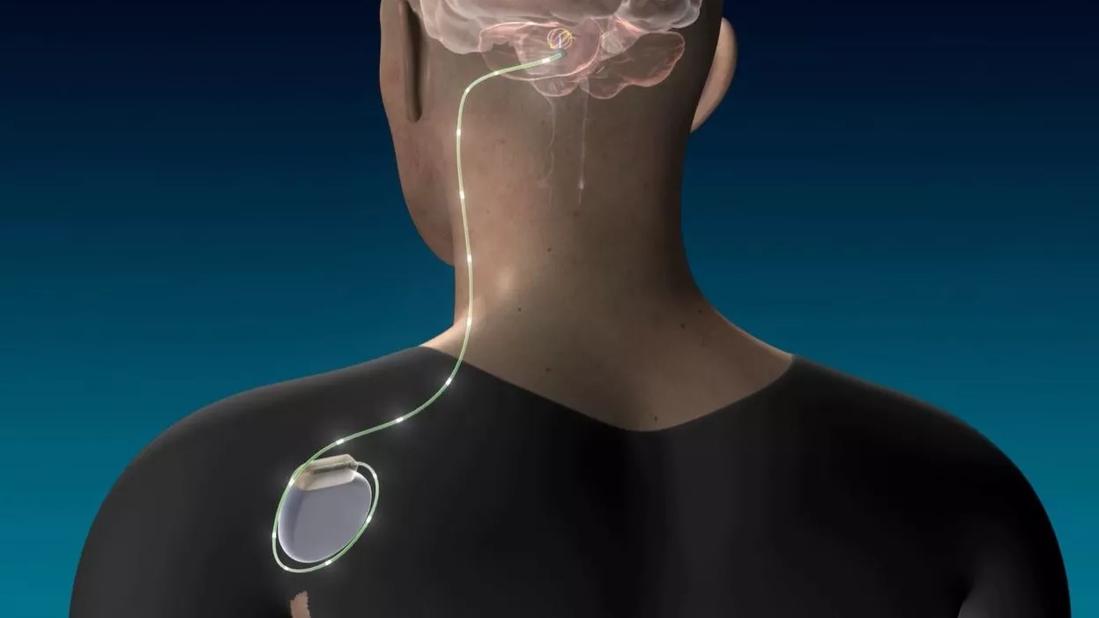
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/89ee648d-08c5-4a1a-97c6-77a61b1c9995/dbs1-jpg)
18-NEU-1184-DBS-Stroke-650×450
Cleveland Clinic researchers have received a $2.5 million grant from the National Institutes of Health (NIH) to further advance their unprecedented use of deep brain stimulation (DBS) of the dentate nucleus to enhance motor rehabilitation in stroke survivors.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The award supports continuing work in preclinical models to improve the efficacy and better elucidate the mechanisms of DBS in restoring post-stroke motor function. It will operate in parallel with the Cleveland Clinic researchers’ ongoing first-in-human clinical trial exploring the use of DBS to restore motor function in patients after a stroke (see previous Consult QD coverage of that trial).
“If this research succeeds, it will offer new hope for patients who have suffered a stroke and remained paralyzed,” says Andre Machado, MD, PhD, Chairman of Cleveland Clinic’s Neurological Institute and co-primary investigator on the grant. “It’s an opportunity to allow patients to rehabilitate and gain function — and thereby gain independence.”
“Our working hypothesis is that low-frequency dentate nucleus DBS augments excitatory dentatothalamocortical output, and in the process increases cerebral cortical excitability, facilitating functional reorganization in perilesional cortical areas and further supporting motor recovery,” adds the other co-primary investigator, Ken Baker, PhD, of the Department of Neurosciences in Cleveland Clinic Lerner Research Institute.
“The experiments proposed in the study to be funded by this grant will help ensure successful human translation of this promising novel treatment while also systematically examining its mechanistic underpinnings,” notes Dr. Machado, a neurosurgeon who is placing the DBS devices into patients participating in the study’s clinical trial component.
Advertisement
The experiments are specifically designed to determine the following:
“There are currently no effective treatments for the hundreds of thousands of individuals who live with chronic motor disabilities following a stroke,” observes Dr. Baker. “This NIH award will help us further refine our work to use cerebellar stimulation in an effort to reduce stroke’s burden on patients, their families and society.”
Advertisement
Advertisement
First full characterization of kidney microbiome unlocks potential to prevent kidney stones
Researchers identify potential path to retaining chemo sensitivity
Large-scale joint study links elevated TMAO blood levels and chronic kidney disease risk over time
Investigators are developing a deep learning model to predict health outcomes in ICUs.
Preclinical work promises large-scale data with minimal bias to inform development of clinical tests
Cleveland Clinic researchers pursue answers on basic science and clinical fronts
Study suggests sex-specific pathways show potential for sex-specific therapeutic approaches
Cleveland Clinic launches Quantum Innovation Catalyzer Program to help start-up companies access advanced research technology