Tag debug info: client: {"assets":{},"datasets":{},"live":{},"projects":{},"users":{},"observable":{"assets":{},"datasets":{},"live":{},"projects":{},"users":{}}} Now: 1770491724295 Cache Key: cqdTagPageBySlug:deep-brain-stimulation-dbs fetchCache[cqdTagPageBySlug:deep-brain-stimulation-dbs].expirationTime: falsey fetchCache[cqdTagPageBySlug:deep-brain-stimulation-dbs]. seconds remaining: falsey All fetchCache expiration times: -- Key: cqdNotFoundPage, seconds remaining: 1025 -- Key: cqdPostsByTag:cqd-migrated-tag-26469,1,10, seconds remaining: -4021 -- Key: cqdTagPageBySlug:sln360, seconds remaining: -3839 -- Key: cqdPostsByTag:cqd-migrated-tag-24541,1,10, seconds remaining: -3772 -- Key: cqdTagPageBySlug:gender-equity, seconds remaining: -2346 -- Key: cqdPostsByTag:cqd-migrated-tag-22582,1,10, seconds remaining: -2304 -- Key: cqdTagPageBySlug:video-eeg, seconds remaining: -2147 -- Key: cqdPostsByTag:cqd-migrated-tag-24971,1,10, seconds remaining: -2090 -- Key: cqdTagPageBySlug:sleep-latency, seconds remaining: 1025 -- Key: cqdPostsByTag:cqd-migrated-tag-26830,1,10, seconds remaining: 1072 -- Key: cqdTagPageBySlug:resilience, seconds remaining: 1898 -- Key: cqdPostsByTag:cqd-migrated-tag-17130,1,10, seconds remaining: 1948 -- Key: cqdTagPageBySlug:khalid-al-muti, seconds remaining: 5233 -- Key: cqdPostsByTag:cqd-migrated-tag-4752,1,10, seconds remaining: 5282 -- Key: cqdTagPageBySlug:hearing-disorders, seconds remaining: 5968 -- Key: cqdPostsByTag:cqd-migrated-tag-1744,1,10, seconds remaining: 6020 -- Key: cqdTagPageBySlug:hyman, seconds remaining: 6178 -- Key: cqdPostsByTag:cqd-migrated-tag-17220,1,10, seconds remaining: 6239 -- Key: cqdTagPageBySlug:stop-bang, seconds remaining: 6381 -- Key: cqdPostsByTag:cqd-migrated-tag-4745,1,10, seconds remaining: 6429 -- Key: cqdTagPageBySlug:debulking-surgery, seconds remaining: 6980 -- Key: cqdPostsByTag:cqd-migrated-tag-20449,1,10, seconds remaining: 7040 -- Key: cqdTagPageBySlug:iridectomy, seconds remaining: 7282 -- Key: cqdPostsByTag:cqd-migrated-tag-22823,1,10, seconds remaining: 7376 -- Key: cqdTagPageBySlug:mona-gupta, seconds remaining: 8203 -- Key: cqdPostsByTag:cqd-migrated-tag-863,1,10, seconds remaining: 8257 conditions: -- false, -- NA, -- NA, -- NA -- false Cache miss for key cqdTagPageBySlug:deep-brain-stimulation-dbs - retrieving from Sanity CCCache.dataFetchCount: 13654 Cache cleanup seconds remaining: 25968
Advertisement
Advertisement
Tag: deep brain stimulation (DBS)
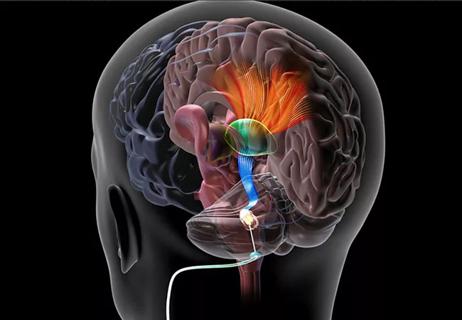
Rapid innovation is shaping the deep brain stimulation landscape
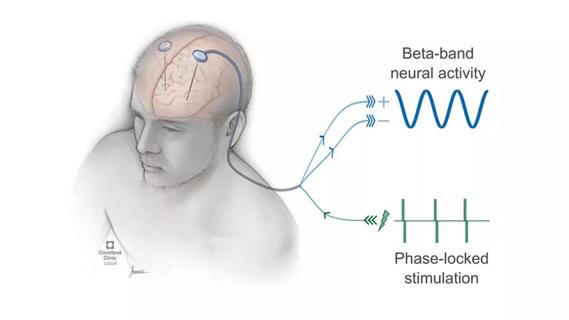
Real-time adjustments may help reduce bothersome dyskinesias
Promising preclinical research indicates functional motor recovery is durable
$3.2 million grant will fund use of calcium-based imaging to record neuronal activity in ischemia model
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
EDEN analysis investigates electrophysiological changes after continuous dentate nucleus stimulation
Research project will leverage insights into neural circuits to advance DBS technology
Study reveals heavy care burden, missed opportunities in patients with uncontrolled symptoms on oral therapy
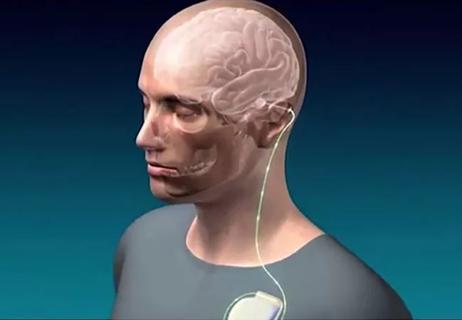
First-in-human trial demonstrates safety, encouraging functional response in chronic phases of recovery
Also will assess ability of high-intensity exercise to offset the cognitive decline
Novel protocol combines gait testing during stimulation with neural signal measurement
Rendered: Sat Feb 07 2026 19:15:24 GMT+0000 (Coordinated Universal Time)
9500 Euclid Avenue, Cleveland, Ohio 44195 |
800.223.2273 | ©
2026 Cleveland Clinic. All Rights Reserved.