The drug does not provide lasting disease control for most patients
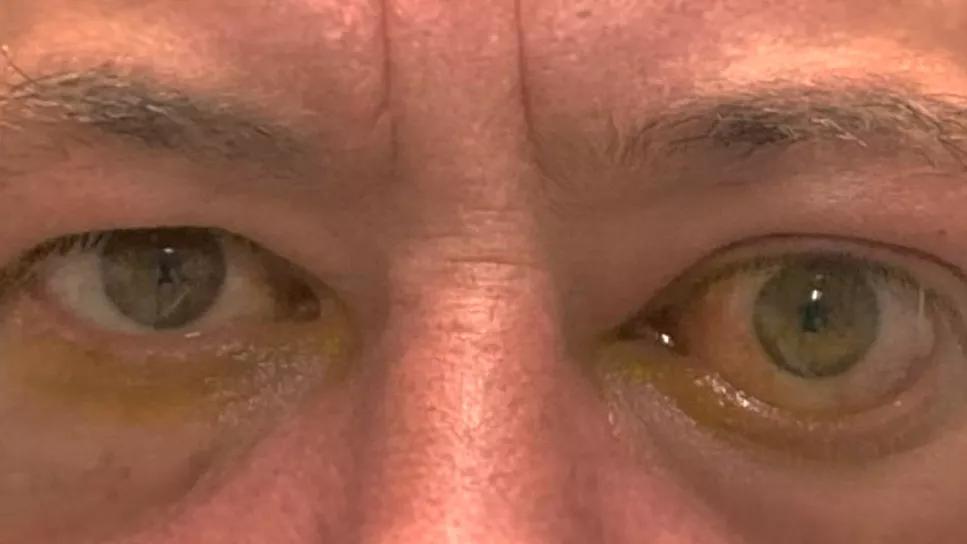
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/704eab52-a442-4c04-a1e6-e7ba139745dd/teprotumumab-to-treat-thyroid-eye-disease)
Thyroid eye disease
This is a reprint of an editorial from the November 2025 issue of Cleveland Clinic Journal of Medicine (2025 Nov 3;92(11):702-703).
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The introduction of teprotumumab as a medical therapy for thyroid eye disease (TED) represented a major advancement in our ability to help patients with this challenging condition. By targeting the insulin-like growth factor 1 receptor (IGF-1R), and thus blocking the thyroid-stimulating hormone receptor and IGF-1R complex expressed by orbital fibroblasts, it became the first biologic aimed specifically at treating TED. Early clinical trials showed impressive proptosis reduction (≥ 3 mm) and improvements in clinical activity score (0 or 1 in most patients),1,2 leading to rapid U.S. Food and Drug Administration approval in 2020.3
Unfortunately, five years into clinical practice, a sobering reality has emerged — not from long-term randomized clinical trials studying durability, because no such trials have been performed, but from real-world longitudinal data with extended follow-up that have revealed teprotumumab does not provide lasting disease control for a large majority of patients.3 While it does modulate TED signs and symptoms temporarily, it does not appear to alter the natural disease course in most patients.
Our recent retrospective analysis offers some of the longest follow-up data on patients treated with teprotumumab.3 The initial proptosis response rate in 21 patients was 84% but decreased to 57% at one year, mirroring the results from the OPTIC-X (Treatment of Graves’ Orbitopathy to Reduce Proptosis With Teprotumumab Infusions in an Open-Label Clinical Extension Study) trial.4 Unfortunately, the failures did not cease at one year. They continued through the entire two years of follow-up in our study.3 Ultimately, only about one-third of patients maintained a sustained response without recurrence.
Advertisement
The relapses weren’t mild setbacks. Most patients experienced reactivation of inflammation (measured by clinical activity score regression) and complete loss of proptosis improvement, with levels often returning to baseline.3 More concerningly, these late flares occurred past the traditional disease activity timeline, suggesting the active phase of TED could be prolonged in patients receiving teprotumumab. Because of these very late flares, the point at which to consider surgery during TED’s stable phase is less reliable. In a condition where reoperation is higher risk, this is a significant clinical conundrum.
The evidence that teprotumumab does not provide long-term improvement for many patients aligns with the concept proposed by North et al5 of modulators, treatments that suppress disease symptoms, and modifiers, treatments that alter disease course. Understanding this conceptual framework, and how teprotumumab fits into it by mainly modulating disease activity temporarily, is essential for managing patient expectations and informing clinical decisions. While modulation has value, the high rates of relapse and late reactivation highlight the need for medical therapies that demonstrate a more durable benefit to patients.
Interest in treating patients who were nonresponders or experienced flares with a second course of teprotumumab has grown. However, adverse effects such as ototoxicity (e.g., tinnitus, hearing loss, ear pain, vertigo) may be significant, and they are directly related to disruption of IGF-1R pathways.6 This can lead to sensorineural hearing loss in adults because IGF-1R is expressed on outer hair cells in the cochlea. Existing data on retreatment are sparse,4,5 but even the small number of published cases have shown potentially severe side effects that led patients to discontinue teprotumumab.4
Advertisement
Despite these challenges, teprotumumab remains an important tool in the TED treatment landscape. Its future likely lies in careful patient selection and in combination therapies. For example, some case reports describe promising results treating mild or early dysthyroid compressive optic neuropathy with teprotumumab,7 though time to improvement onset and variable response to treatment limit use in urgent cases.
An ongoing postmarketing study is assessing different dosing regimens, with some participants receiving up to 16 infusions.8 However, the one-year follow-up period falls short, given that recurrences can occur up to two years after treatment.
Partial treatment (< 8 infusions) also seems to provide good results,9 so administering doses over an extended period may potentially overcome some of teprotumumab’s limitations, but definitive guidance will depend on the results of future studies.
More promising is the idea of combination therapy with agents such as tocilizumab, which targets interleukin 6, to possibly provide a longer lasting immunosuppressive effect alongside IGF-1R blockade.10 Similarly, orbital radiotherapy, previously de-emphasized as a treatment for TED,11 may merit reexploration as a fall-back option or a co-modality that could help mitigate inflammatory flares after teprotumumab therapy. We may be entering an era where teprotumumab represents one component of a larger, multimodal disease management strategy.
The enthusiasm for teprotumumab is well warranted. A biologic targeting TED that had compelling early results filled a major therapeutic gap. But real-world durability falls short for two-thirds of treated patients, and they experience disease flares long after TED onset, when many untreated patients may not. These findings, coupled with teprotumumab’s side effects and immense cost, temper that initial enthusiasm.
Advertisement
Like many promising medical therapies, the role of teprotumumab in treating TED will evolve. It will likely find its place — not as a cure-all but as a modulating agent used thoughtfully and often in combination with other treatments. Perhaps in concert with other therapies, teprotumumab can better address the full clinical arc of TED.
Dr. Perry is an oculofacial plastic surgeon at Cleveland Clinic Cole Eye Institute.
References
Advertisement
Advertisement
A review of pathophysiology, risk factors, clinical presentation and treatment options
Registry data highlight visual gains in patients with legal blindness
Study is first to show reduction in autoimmune disease with the common diabetes and obesity drugs
It’s the first step toward reliable screening with your smartphone
Fixational eye movement is similar in left and right eyes of people with normal vision
Oral medication may have potential to preserve vision and shrink tumors prior to surgery or radiation
A new online calculator can determine probability of melanoma
Only 33% of patients have long-term improvement after treatment