Despite significant progress, questions remain
By Rishi P. Singh, MD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
(Dr. Singh, of Cleveland Clinic Cole Eye Institute, received the 2018 early career investigator award from the president of the American Society of Retina Specialists. He authored 15 peer-reviewed publications and presented at 18 meetings on diabetic eye disease in 2018. In this article, he highlights research conducted at Cole Eye Institute in 2017.)
Diabetic retinal disease is the leading cause of blindness in working-age adults in the United States. Over the past few years, there have been significant advancements of our understanding of how this disease develops, what the risk factors are for progression and how to treat both diabetic macular edema (DME) and diabetic retinopathy (DR). We are fortunate to have advances in imaging such as widefield angiography and optical coherence (OCT) angiography for disease detection. In addition, we have multiple intravitreous antivascular endothelial growth factor drugs (anti-VEGF) available for treatment.
Despite this, there are multiple unanswered questions about the disease pathogenesis, response to treatment and how we prevent complications. This a major thrust of research at Cole Eye Institute.
There have been studies of human pathological specimens demonstrating significant capillary loss within the choroid, especially in advanced diabetic retinopathy (Figure 1). But what about in vivo? We assessed choroidal capillary density by OCT angiography and found significant loss of the choriocapillaris vessels in patients with advanced diabetic retinopathy. As a result, one might also consider the choroid to be a contributor of ischemia seen in diabetic retinopathy and to the development of proliferative diabetic retinopathy.
Advertisement
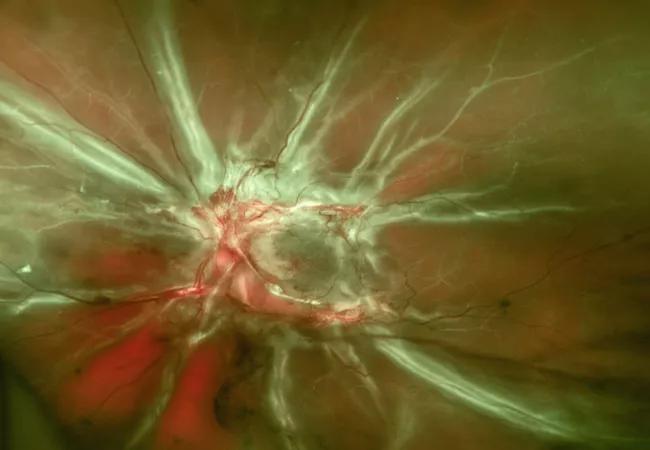
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/8bcd47b9-c4b6-484e-8cd5-7245a74ed997/18-EYE-1206-Singh-CQD-1_jpg)
Figure 1. Retinal fundus color photograph demonstrating advanced proliferative diabetic retinopathy.
Previous studies such as the Diabetes Control and Complications Trial (DCCT) have shown that intensive therapy reduced long-term complications. However, strict control in the first two years resulted in a higher rate of proliferative retinopathy. We investigated this further in a population of patients who underwent bariatric surgery for diabetes. These patients experienced a 3 percent drop in hemoglobin A1c (HbA1c) on average from baseline. When looking at microvascular outcomes, there was no difference seen in the bariatric surgery treated group and the intensive medical management group. Thus, patients who undergo bariatric surgery can be assured that ocular complications in the first few years are unlikely despite large drops in their blood sugar.
We investigated whether baseline metabolic factors affected anti-VEGF mediated improvements in patients with DME and DR. For both diseases, none of the systemic factors affected anti-VEGF treatment outcomes, including systemic characteristics, glycemic control and renal function (Figure 2).
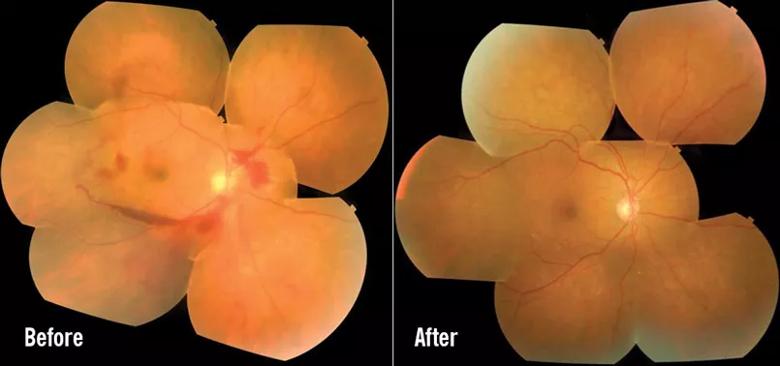
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/a841b644-08a5-4093-bd37-e92ba09a4607/18-EYE-1206-Singh-CQD-2_jpg)
Figure 2. Retinal fundus color photograph showing improvements in DR outcome following anti-VEGF treatment.
One of the more interesting findings concerned the outcomes of those who received focal laser treatment. There was a clear inverse relationship between higher baseline HbA1c levels and visual acuity improvement with focal laser, with no improvement seen in the highest quartile. This should give clinicians pause when considering focal laser treatment, especially when a patient has uncontrolled diabetes.
Advertisement
Cataract surgery is a common complication of diabetic eye disease and is the most common outpatient eye surgery performed. However, patients with diabetic retinopathy in particular are at high risk of developing macular edema following even uncomplicated cataract surgery. How can we prevent this complication in this at-risk patient population?
In two prospective randomized studies, we demonstrated that the addition of a nonsteroidal agent to the postoperative regimen can decrease the rates of macular edema from 15.9 percent to 4.1 percent and increase those achieving a 3-line gain in vision following cataract surgery by 19 percent over the standard of care regimen. Patients, especially those with baseline diabetic retinopathy, can achieve better anatomic and visual outcomes following cataract surgery with the addition of a nonsteroidal agent during the postoperative period.
We are excited to establish the Cole Eye Institute Center for Excellence in Ophthalmic Bioinformatics, which will address leading causes of blindness, including age-related macular degeneration, diabetic eye disease, retinal vein occlusions, glaucoma and uveitis. Understanding presentations of these ophthalmic diseases, their risk factors, their prognosis for progression and their optimal response to treatment through the bioinformatics processes is an important step at improving the lives of our patients.
Advertisement
Advertisement

Motion-tracking Brillouin microscopy pinpoints corneal weakness in the anterior stroma

Registry data highlight visual gains in patients with legal blindness

Prescribing eye drops is complicated by unknown risk of fetotoxicity and lack of clinical evidence

A look at emerging technology shaping retina surgery

A primer on MIGS methods and devices

7 keys to success for comprehensive ophthalmologists

Study is first to show reduction in autoimmune disease with the common diabetes and obesity drugs

Treatment options range from tetracycline injections to fat repositioning and cheek lift