General principles for use of the long-awaited new therapy approach
After more than a decade of clinical testing and development, the first two renal denervation systems for treatment of uncontrolled hypertension gained U.S. marketing clearance in November, and Cleveland Clinic was among the first in the nation to use them in patients outside of a clinical trial setting.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
In early November, Cleveland Clinic was one of the first four U.S. centers to use the Paradise™ ultrasound renal denervation system (Recor Medical) following its Nov. 7 FDA approval. Later that month, a Cleveland Clinic team was among the first in the U.S. to use the Symplicity Spyral™ renal denervation system (Medtronic) in the wake of its Nov. 17 FDA approval. In both cases, Cleveland Clinic had patients with resistant hypertension waiting in the wings for approval of the technologies for commercial use.
“Renal denervation is a unique opportunity to treat patients with hypertension, which is a silent killer,” says Aravinda Nanjundappa, MBBS, MD, the interventional cardiologist on both cases. “It can really be a game changer in terms of lowering cardiovascular morbidity and mortality.”
For hypertension specialists like Luke Laffin, MD, Co-Director of Cleveland Clinic’s Center for Blood Pressure Disorders, FDA approval of the technology was well received. “Renal denervation represents an innovation in a disease space that has had limited innovations for many years,” he says. “It represents an entirely different treatment from the everyday pill that most of us are used to when discussing hypertension treatment.”
Prior to joining Cleveland Clinic, Dr. Nandunjappa gained firsthand experience with renal denervation through his participation in a clinical trial of the Symplicity system. “It’s a straightforward procedure similar to renal angioplasty,” he says.
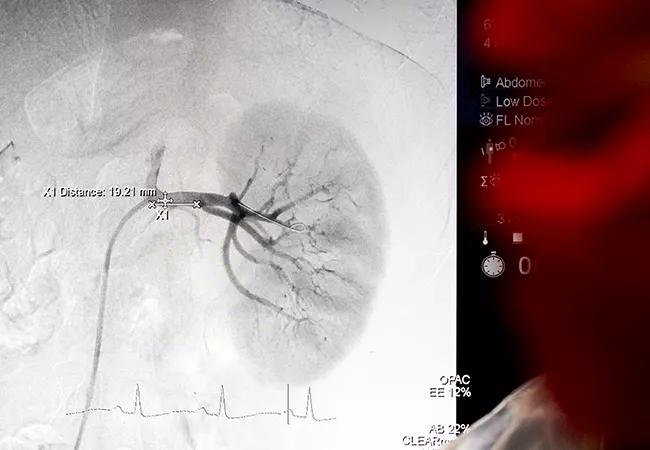
Renal denervation requires inserting a catheter in the femoral artery and taking a renal angiogram to visualize the renal arteries. A balloon catheter is then inserted in the arteries and ultrasound (Paradise) or radiofrequency energy (Symplicity) is delivered through the entire renal arterial system.
Advertisement
“You have to ablate all the nerves,” Dr. Nanjundappa notes. “They usually start at the proximal portion of the artery and go all the way to the kidney parenchyma.”
Both of the initial cases done at Cleveland Clinic in November were performed using conscious sedation with careful attention to pain management. Both were successful and well tolerated, with no challenges or complications.
The Paradise procedure was completed in less than an hour. Because it was done late in the evening, the patient was kept overnight. “Most patients can be sent home the same day,” Dr. Nanjundappa says.
The Symplicity procedure — the first in Ohio following the system’s FDA approval — involved use of a single catheter and completion of denervation of both renal arteries in less than an hour.
Dr. Nanjundappa reviewed and discussed the index cases prior to renal denervation in a multidisciplinary fashion with Dr. Laffin from preventive cardiology, Scott Cameron, MD, PhD, from vascular medicine and Khaled Ziada, MD, from interventional cardiology. He also credits the cardiac catheterization lab team of nurses, technicians and ancillary staff as instrumental to the success of renal denervation procedures.
Renal denervation has been done for years in the context of clinical trials and appears to be safe in both the short and long term. “No increased risk of hypotension or adverse responses, such as renal artery stenosis, has been seen,” Dr. Laffin notes.
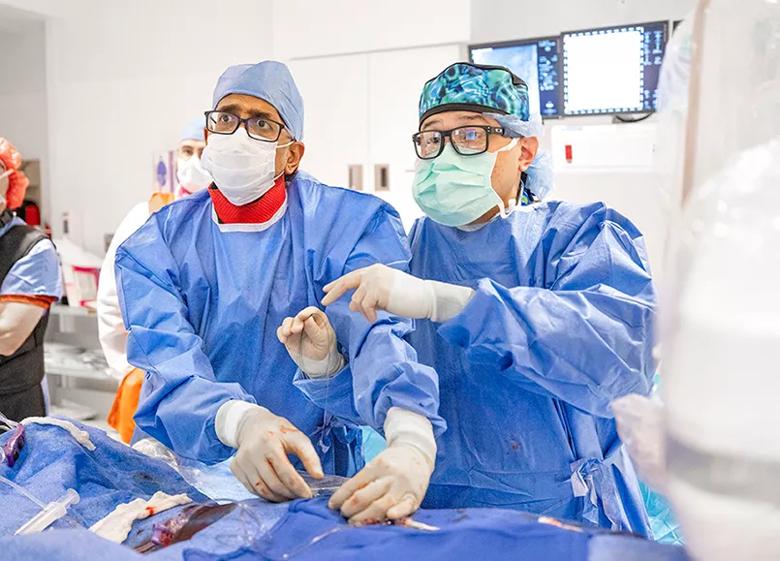
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/4cca75c2-73b0-4649-8076-7c7b192ef6d7/23-HVI-4401682-CQD-Inset1_jpg)
Dr. Nanjundappa (left) and fellow Calvin Sheng, MD, in the cath lab during Cleveland Clinic’s first post-approval case using the Paradise renal denervation system.
Advertisement
Renal denervation lowers systolic blood pressure by an average of 5 to 6 mmHg — about the equivalent of a single antihypertensive medication. The effect appears to be durable: data with the Symplicity system show the drop in blood pressure sustained at five years; data with the Paradise system are comparable but currently extend to only 12 months.
The clinical trials of both technologies included two types of patients: those with resistant hypertension, and those with hypertension who were medication-free for one month prior to and several months following the procedure. Renal denervation was effective in both.
“Some data show that lower-risk patients — for example, those on one or two antihypertensive medications — could also benefit from it,” Dr. Laffin says, adding that Cleveland Clinic will not initially be using renal denervation for that patient population. “The vast majority of patients we plan to treat will have resistant hypertension or, less commonly, be intolerant of many classes of antihypertensive medications.”
With plenty of patients worldwide who fit those categories, interest in renal denervation is high.
Initially, the procedure will be done predominantly at major medical centers or those that were involved in renal denervation clinical trials, Dr. Laffin expects, but everyone will be watching the real-world outcomes. “If and when we reproduce the results from the trials, the availability of the procedure at smaller centers will expand,” he predicts.
Both physicians recommend that a hypertension specialist serve as gatekeeper to determine a patient’s suitability for renal denervation and make appropriate referrals.
Advertisement
“Our patients first have to see a hypertension specialist in our Center for Blood Pressure Disorders,” Dr. Laffin explains. “If interested patients meet criteria that our specialists have agreed upon, we will do the appropriate testing — typically, assessing their renal arteries noninvasively — and then get them set up with an interventional cardiologist for a date in the cath lab.”
According to Dr. Nanjundappa, renal denervation should be performed by specialists with training in peripheral intervention. “This is important for the patient’s safety,” he says.
Cleveland Clinic plans to use both FDA-approved renal denervation systems. “We are device-agnostic,” says Dr. Laffin, who notes that insurance coverage for both systems has yet to be finalized.
After patients undergo renal denervation, Cleveland Clinic will leave most patients on their antihypertensive medication regimen and reassess their blood pressure at one month. “It should help most people get their blood pressure into a better range,” Dr. Laffin says. “If the blood pressure goes too low, we can always pull back on their medications.”
At this time, there is no way to know who will respond to renal denervation and who will not. That is not a reason to withhold the procedure, Dr. Laffin notes.
“In patients whose hypertension is still uncontrolled on three, four or five medications, or who cannot or will not take another pill, I think this is a reasonable option, given the safety of this procedure and the potential benefit,” he says.
Advertisement
“In nearly everything we do at Cleveland Clinic, teamwork and partnership is imperative in bringing the best care to patients,” says Amar Krishnaswamy, MD, Section Head of Interventional Cardiology, who has previous research experience with renal denervation and helped bring the technology to Cleveland Clinic. “In the case of renal denervation, our group in interventional cardiology, working with colleagues across cardiovascular medicine and nephrology, is pleased to have the opportunity to bring this newly approved technology to hypertensive patients who stand to benefit.”
Advertisement

Fixed-dose single-pill combinations and future therapies

Clinicians should individualize dosing practices based on patient risk factors and preferences

Results inform dose selection for further trials of the aldosterone synthase inhibitor

Phase 2b trial of aldosterone synthase inhibitor also finds no benefit from dose escalation

More than 30% of patients die when early dialysis is needed after surgery
Pioneering and refining the approach in pyeloplasty, nephrectomy and more
Fully-automated process uses preop CT, baseline GFR to estimate post-nephrectomy renal function
New protocol reduces costs, increases patient and caregiver satisfaction