Resulting localization of a single focus paves way for laser ablation
By Imad Najm, MD, and Dileep Nair, MD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
A 35-year-old right-handed woman was referred to Cleveland Clinic’s Epilepsy Center for possible surgical management of her pharmacoresistant epilepsy. Her seizures had started at age 31. She had failed multiple antiepileptic medications (total of six) and had an MRI of the brain showing multiple periventricular nodules (Figure 1).
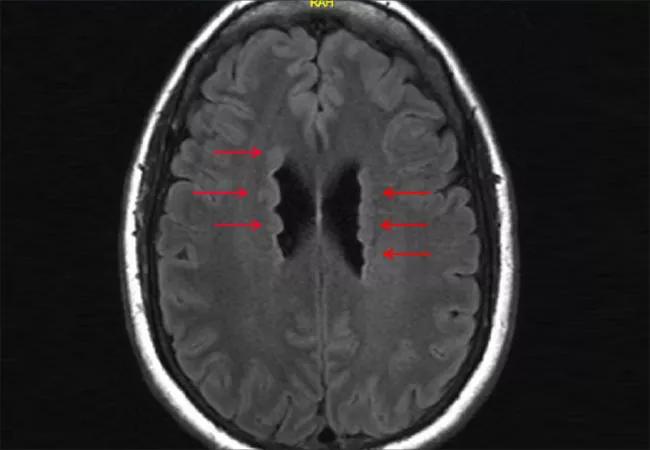
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/61e9db38-6f6f-482f-9dd6-16c7f9e180f1/18-NEU-4011-najmNair-1-650x450_jpg)
Figure 1. Brain MRI prior to the patient’s presentation at Cleveland Clinic showing multiple periventricular nodules (arrows).
The patient described multiple seizure types that included staring with unresponsiveness, difficulty finding words, euphoric feelings and episodes of muffled hearing followed by blurry vision that at times evolved into secondary generalized tonic-clonic convulsions.
She underwent a complete noninvasive evaluation that included video EEG monitoring, 3-tesla MRI, FDG-PET scan, MEG and ictal SPECT. As detailed in Table 1, these tests revealed multiple areas of possible epileptic abnormalities, with the right hemisphere affected more than the left.
On the basis of these results, the multidisciplinary team in our patient management conference recommended a stereoelectroencephalography (SEEG) evaluation with extensive coverage of the right hemisphere:
The SEEG evaluation revealed interictal spikes and ictal patterns that were distributed to one of the right nodules, the right posterior perisylvian region and the right lateral parietal area (Figure 2).
Advertisement
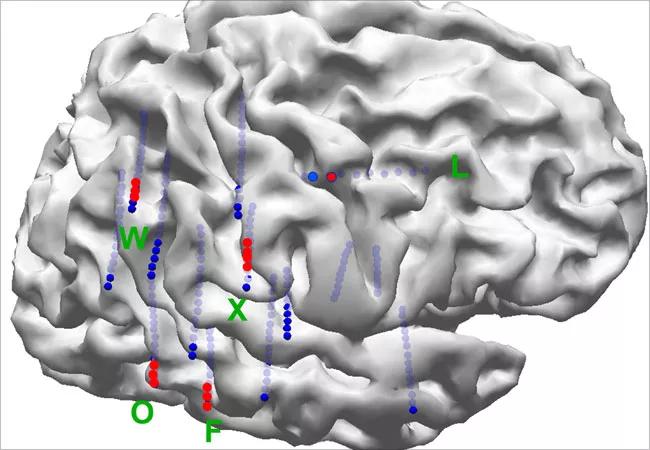
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/1339d2f8-25d1-42f7-a6af-b23907ff60b9/18-NEU-4011-najmNair-2-650x450_jpg)
Figure 2. SEEG implantation map showing distribution of interictal spikes and ictal patterns in one of the right nodules, the right posterior perisylvian region and the right parietal area.
These findings suggested two main possibilities: (1) The epileptic activities are multifocal. (2) The epileptic activities are generated in a single area — perhaps one of the periventricular nodules — with spread to the other areas. Surgical intervention would be entertained only if the second hypothesis was confirmed.
To test this hypothesis, we used the cortico-cortical evoked potentials (CCEPs) technique that was developed at Cleveland Clinic. This technique allows for the testing of connections between various areas in the brain and the direction(s) of those connections. The electrodes that showed epileptic activities were stimulated. CCEPs were elicited in most of the cortical areas with epileptic activity after stimulation of the epileptic periventricular nodule (Figure 3).
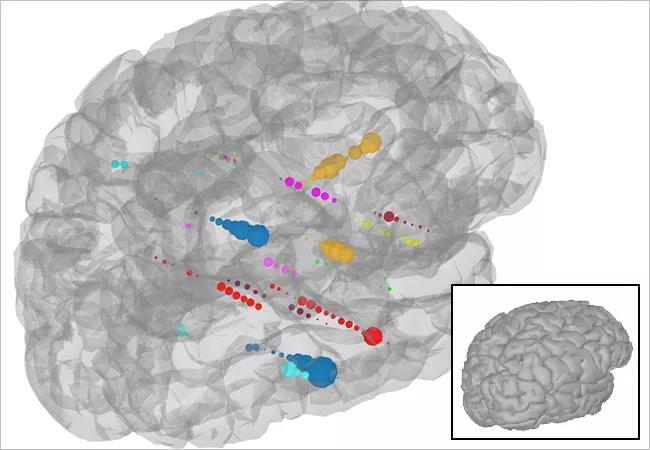
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/329d258d-9ede-4ae8-9a4e-a52b50b1bc41/18-NEU-4011-najmNair-3-650x450_jpg)
Figure 3. Illustration of the connectivity of the surrounding cortex from a seed point in the periventricular nodule that was involved in the ictal onset. Electrode stimulation from the periventricular nodule at low frequencies generates a measure of effective brain connectivity known as cortico-cortical evoked potentials (CCEPs). The regions of greatest connectivity are depicted as larger-sized recording electrodes. This methodology has shown the ability to map the propagation network of seizures. It suggests that the extent of the propagation network is quite broad, which could explain how a relatively restricted epileptogenic zone in the periventricular nodules could give rise to early propagated ictal activity in various parts of the lateral parietal and temporal neocortex.
Advertisement
Figure 4 shows representative CCEP responses from the patient. The top panel shows an accentuated response from some of the electrodes involved in the ictal activity (electrodes from L, W and X). We believe these accentuated CCEP responses arise in brain regions capable of generating synchronous brain activity, which is one of the hallmarks of epilepsy. The bottom panel highlights the CCEP response from stimulating the electrodes situated in the periventricular nodular heterotopia. The color pattern (pink, light blue and light orange) along the largest peaks of this CCEP response shows distribution of response to the posterior perisylvian and right lateral parietal regions shown in the accompanying segmented brain image. This suggests that the periventricular nodule may be the primary generator of the epilepsy and its connections to the outer surface of the brain, explaining the rapid spread of epileptic activity.
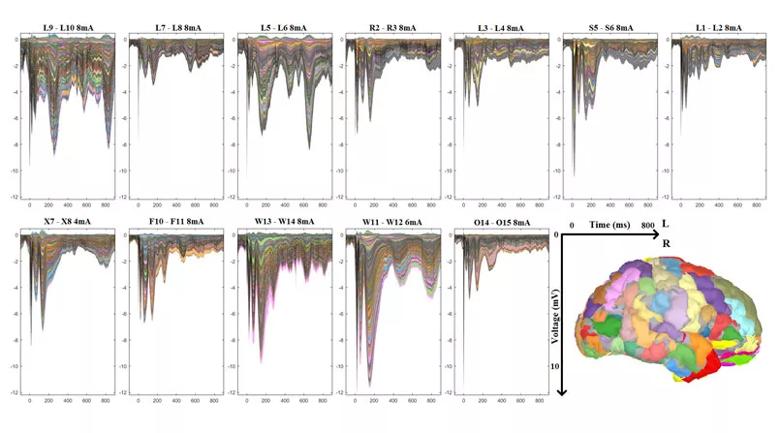
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/f49ba798-efff-497d-a2df-15084a351c47/18-NEU-4011-najmNair-4Top-805x447_jpg)
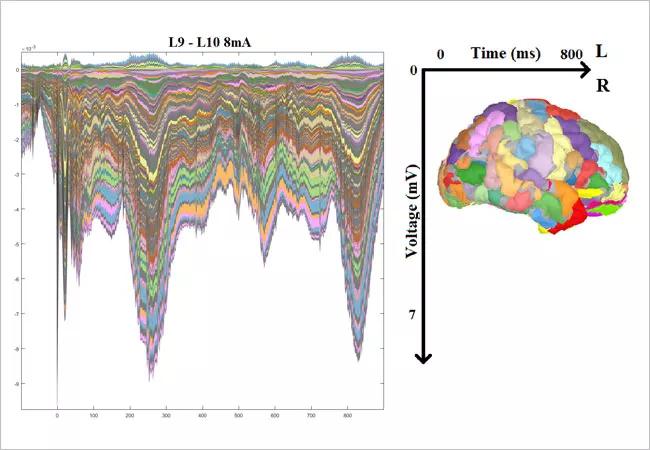
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/84b6c25e-393d-4721-b861-6932130ffcf9/18-NEU-4011-najmNair-4Btm-650x450_jpg)
Figure 4. CCEP responses from the patient. The top panel shows an accentuated response from some of the electrodes involved in the ictal activity (electrodes from L, W and X). The bottom panel highlights the CCEP response from stimulating the electrodes situated in the periventricular nodular heterotopia. See main text for a detailed interpretation.
These results suggested that the patient had right hemisphere focal epilepsy that probably originated from one of the right lateral ventricle nodules, activating the inferior parietal network. However, independent foci between the two regions cannot be completely ruled out.
Advertisement
The patient underwent laser ablation of the right lateral ventricle epileptogenic nodule (see Figure 5 for post-ablation imaging), and she has remained seizure-free since 2015.
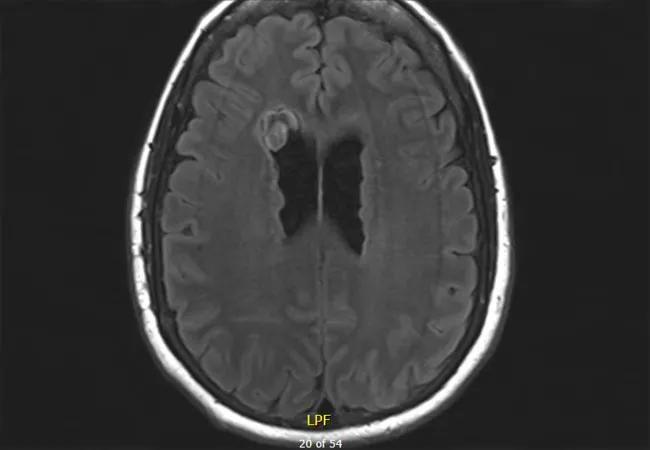
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/f23a62f4-7fc2-40b7-8efa-02ed33d8149b/18-NEU-4011-najmNair-5-650x450_jpg)
Figure 5. MRI taken following laser ablation of the periventricular nodules determined to be involved in the patient’s seizure generation. Note the focal changes (compared with the pre-intervention MRI in Figure 1) due to the ablation.
This case illustrates the power of using a state-of-the-art multimodality evaluation in patients suffering from pharmacoresistant epilepsy in the setting of periventricular nodular heterotopias. The SEEG method enables the sampling of both deep (periventricular nodules) and cortical areas in a single setting. The use of CCEPs to study the connectivity between various brain areas and the directions of those connections helps in uncovering the possibility that there may be a single pacemaker (epileptogenic focus) connected to multiple areas. In turn, the resulting identification and localization of a single focus allows the use of the laser technique for ablation of the deep focus, leading to a higher chance of complete seizure control with low surgical morbidity.
Dr. Najm is Director of Cleveland Clinic’s Epilepsy Center. Dr. Nair is Section Head of Adult Epilepsy and Director of Intraoperative Neurophysiologic Monitoring in the Epilepsy Center.
Advertisement
Advertisement

Aim is for use with clinician oversight to make screening safer and more efficient

Rapid innovation is shaping the deep brain stimulation landscape

Study shows short-term behavioral training can yield objective and subjective gains

How we’re efficiently educating patients and care partners about treatment goals, logistics, risks and benefits

An expert’s take on evolving challenges, treatments and responsibilities through early adulthood

Comorbidities and medical complexity underlie far more deaths than SUDEP does

Novel Cleveland Clinic project is fueled by a $1 million NIH grant

Tool helps patients understand when to ask for help