Benefits of novel PET radiotracer expected to go beyond multiple sclerosis
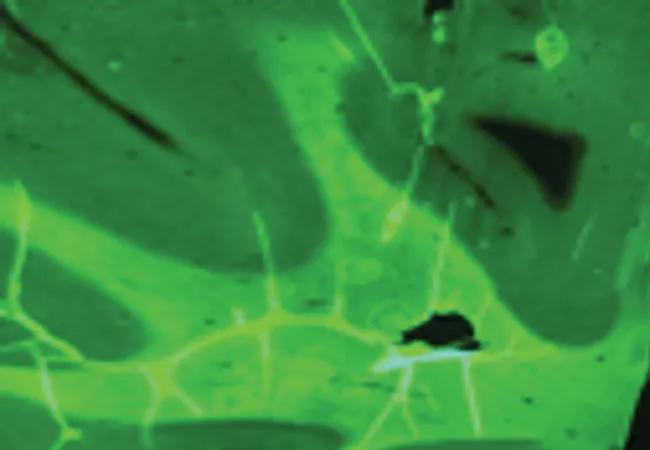
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/9dfa8d31-6713-47ca-9f58-44e177a8208c/19-NEU-3935-myelin-PET-650x450_jpg)
19-NEU-3935-myelin-PET-650×450
A novel approach for accurately characterizing and quantifying myelin in the central nervous system (CNS) has been bolstered with support from a new $1.6 million R01 grant from the National Institutes of Health (NIH).
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
The strategy, based on the development of a positron emission tomography (PET) radiotracer that binds with high affinity to myelin, is anticipated to improve on MRI, which is the current leading method for diagnosing and monitoring progression of multiple sclerosis (MS) but which correlates imperfectly with clinical outcomes.
“This research offers a promising new approach to directly and specifically assess myelin in the brain and spinal cord,” says Robert Fox, MD, Vice Chair for Research in Cleveland Clinic’s Neurological Institute and one of two principal investigators on the grant. “We expect that it will enable clinicians to more unambiguously diagnose MS and monitor disease progression and repair processes.”
Dr. Fox’s fellow principal investigator is Yanming Wang, PhD, of Case Western Reserve University in Cleveland. The myelin PET research initiative is a collaboration between the Neurological and Imaging Institutes at Cleveland Clinic and Case Western Reserve.
Because MRI signal intensity reflects changes in tissue water content, it is a nonspecific measure of macroscopic tissue injuries that are not necessarily caused by myelin damage. Hence, its sensitivity is limited in MS, as is its quantification capability.
To overcome these limitations of MRI, a team of Case Western Reserve researchers developed a series of PET-detectable myelin-imaging agents that penetrate the blood-brain barrier and selectively localize in the brain and spinal cord in proportion to myelin content. The most promising molecule, which will be used in the current investigations, is the radioligand [N-methyl-11C]-4,4’-diaminostilbene (now known as Myeliviz). (For more on this myelin-targeting molecular probe, see this previous Consult QD post.)
Advertisement
Myeliviz has been independently validated and confirmed to directly detect and quantify myelin changes in the CNS with good spatial and temporal resolution, according to Dr. Fox, who is also a staff neurologist in Cleveland Clinic’s Mellen Center for Multiple Sclerosis. The PET image at the top of this post shows robust Myeliviz uptake in normal human brain tissue.
“Myeliviz provides a tool to characterize myelin based on its molecular integrity, which may be useful in the detection of MS lesions earlier than with MRI, and which may also be useful in a broad spectrum of diseases affecting the central nervous system,” notes Mykol Larvie, MD, PhD, Director of Functional Neuroimaging in Cleveland Clinic’s Imaging Institute and a co-investigator on the grant. “This may allow earlier and more accurate diagnosis of MS and may also provide a new approach to evaluating the efficacy of disease-modifying therapies.”
“Uptake of Myeliviz appears to be affected by demyelination and not by inflammation alone, processes that coexist in MS but are indistinguishable by MRI,” adds Dr. Fox.
Myeliviz has previously been tested in rodents and has fulfilled requirements for an FDA Investigational New Drug submission. The NIH grant will enable Cleveland Clinic researchers to further evaluate Myeliviz PET imaging in the laboratory as well as in healthy volunteers.
Specific aims include:
Advertisement
The investigators anticipate that success in these investigations would pave the way for phase 2 clinical trials in patients with MS.
The researchers believe that having a technique to directly and quantitatively evaluate myelin integrity and distribution in the CNS will also mitigate problems vexing MS research. “Being able to accurately assess myelination will be a great help for development and testing of new drugs, especially those based on myelin repair in both relapsing and progressive MS,” says Dr. Fox.
He explains that most current MS therapies — which have not been successful in promoting long-term recovery — target inflammation rather than myelination processes. Evaluation of new cell-based therapies currently in development that target myelination has been hampered by an inability to assess and quantify myelin changes in living patients.
The whole-body and 3D capabilities of PET imaging also offer significant research advantages over postmortem histology, which suffers from potential sampling bias and the inability to conduct quantitative analyses of the entire CNS.
Dr. Fox anticipates that Myeliviz PET will address these unmet needs and also have important implications for other diseases, including use in evaluation of candidates for epilepsy surgery. “Many neurological conditions involve myelin damage or loss, including traumatic brain injury and stroke,” he observes. “We expect that benefits from this new strategy will be widespread.”
Advertisement
Advertisement
First full characterization of kidney microbiome unlocks potential to prevent kidney stones
Researchers identify potential path to retaining chemo sensitivity
Large-scale joint study links elevated TMAO blood levels and chronic kidney disease risk over time
Investigators are developing a deep learning model to predict health outcomes in ICUs.
Preclinical work promises large-scale data with minimal bias to inform development of clinical tests
Cleveland Clinic researchers pursue answers on basic science and clinical fronts
Study suggests sex-specific pathways show potential for sex-specific therapeutic approaches
Cleveland Clinic launches Quantum Innovation Catalyzer Program to help start-up companies access advanced research technology