A long-overdue technology is poised to reshape practice
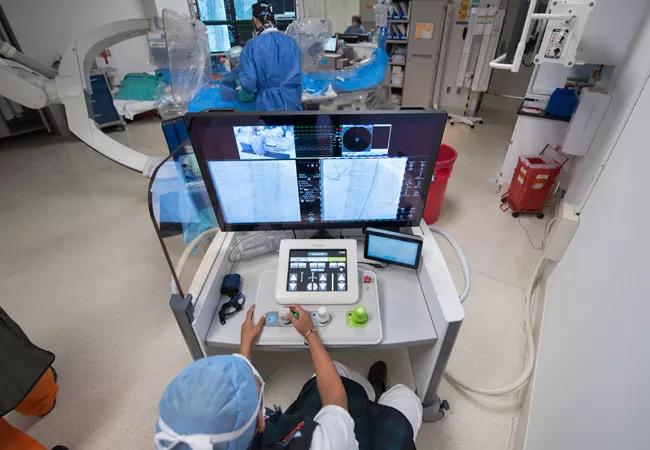
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/e10ee15c-7c13-4893-9547-a2103ec9c3ca/19-HRT-5504-Khatri-CQD-2_jpg)
19-HRT-5504-Khatri-CQD-2
For most providers, the Hippocratic oath’s dictate to “first do no harm” is sacrosanct in its intended application to patient care. Yet providers are not always so diligent about extending this principle to themselves in their professional lives, particularly in the catheterization lab.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“Radiation exposure in the cath lab is what interventional cardiologists tend to worry about most,” says Jaikirshan Khatri, MD, a staff physician in Cleveland Clinic’s Section of Invasive and Interventional Cardiology. “Rates of left eye cataracts and left-side brain tumors within our subspecialty are alarming. The risks from radiation exposure are real.”
And the most widely used means to protect against that exposure — the lead apron — is not without major drawbacks of its own in terms of orthopaedic strain and injuries. “I’m not sure I know an interventional cardiologist who doesn’t have back problems from standing for hours on end with a lead apron on,” notes Dr. Khatri.
These occupational safety concerns are a key reason Cleveland Clinic installed a robotic system — the Corindus CorPath® GRX Vascular Robotic System — in one of its cath labs in June 2019. Cleveland Clinic has used the system for coronary interventions in patients since July, and Dr. Khatri is now using it for about 80% of his caseload.
“The robotic system reduces the operator’s radiation exposure by 95% and liberates him or her from lengthy standing with the lead apron on,” he observes. “No other solution on the market addresses both of these major challenges to operators’ health.”
The robotic system allows the operator to remotely control the equipment essential to percutaneous coronary and peripheral vascular interventions — i.e., guidewire, balloon and stent platforms as well as the guide catheter — from the safety of a radiation-shielded cockpit. This is done via a remote control panel in the cockpit with a joystick and touchscreen controls to guide an articulated robotic arm mounted to the cath lab table. Attached to the robotic arm is a disposable sterile cassette to which the guide catheter is connected and through which intravascular tools are advanced and retracted.
Advertisement
The robotic arm is maneuverable for both radial and femoral access. Notably, however, while the current system is compatible with rapid-exchange designs, its cassette cannot be used with over-the-wire equipment.

Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/b6155609-0ce9-467c-88dd-4e02e0fe9a20/Inset-1_png)
The portions of the robotic system in the procedural theater: Left panel shows the articulated robotic arm mounted to the cath lab table, and right panel shows the sterile cassette attached to the robotic arm.

Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/6b043f9d-f344-4fa5-b962-bab4d9539a9e/Inset-2_png)
A view of the robotic system’s radiation-shielded cockpit on the left, with a close-up on the right of Dr. Khatri operating the cockpit’s remote control panel.
The CorPath GRX is the second-generation robotic system from Corindus, which built on the initial CorPath 200 system by introducing the ability to control the catheter. “That refinement was key to making the technology useable in a wide range of coronary interventions,” says Dr. Khatri.
While other robotic technology systems have been used for endovascular peripheral and aortic vascular procedures, the two CorPath systems are the only ones approved for coronary interventions in the U.S.
Dr. Khatri and his colleague Rishi Puri, MD, PhD, were the first Cleveland Clinic interventional cardiologists to gain proficiency in robotically assisted procedures, but others are expected to be trained soon. These two physicians currently use the robotic system for the full range of coronary interventions, from straightforward to highly complex lesions, including chronic total occlusions (CTOs).
Dr. Khatri had treated about a dozen CTOs with the robotic system within the first two months of using it. “Most CTO cases need to be started manually because they require over-the-wire equipment, but they typically can be completed with the robot,” he says, adding that the same is true of cases involving atherectomy. Likewise, some cases with highly complex coronary anatomy need to be converted to manual procedures near the end because the needed equipment simply cannot be delivered by the robot.
Advertisement
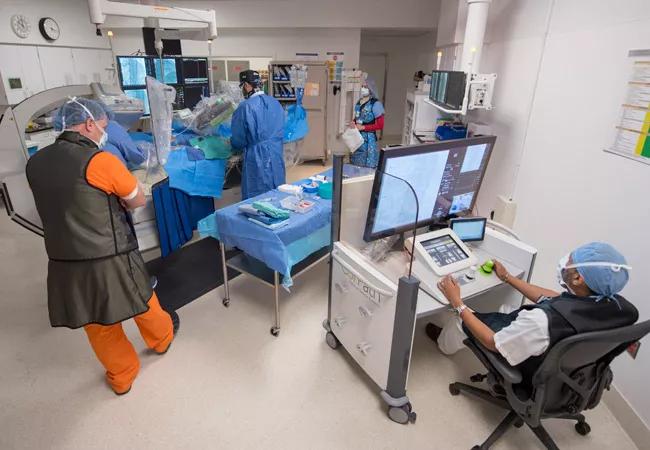
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/02466217-6dd0-4cd0-8e5d-46fd7ef44fae/19-HRT-5504-Khatri-CQD-1_jpg)
Dr. Khatri seated in the cockpit of the robotic system during a case. Although he is shielded from radiation in the cockpit, he may keep his lead apron on throughout a complex case to be ready to return to the table for portions of the case that may need to be done manually because over-the-wire equipment is required.
The ability to handle over-the-wire equipment is the main enhancement that Dr. Khatri expects from the next generation of robotic systems. “That’s the one thing that forces us to plan around the robot,” he explains. “If we could manage over-the-wire equipment, we could do pretty much all cases robotically from start to finish.”
Indeed, being able to weigh in on the next generation of robotic systems was a key impetus for Cleveland Clinic to acquire the robotic system at this time. “The technology isn’t yet quite where it ultimately needs to be, so we wanted to be able to offer our experiences and opinions to shape its future development,” Dr. Khatri says.
To that end, Cleveland Clinic has started a database to collect and evaluate its experience with the technology and share it with developers and the larger interventional cardiology community.
While Cleveland Clinic had not yet used the robotic system for endovascular treatment of peripheral vascular disease as of early autumn 2019, expansion along those lines is anticipated soon.
“The arteries involved in those procedures are typically nice and straight, so it’s even easier for the robot to work well in these cases,” says Dr. Khatri, who also performs peripheral vascular procedures. And because peripheral vascular interventions tend to involve more radiation exposure, he says there’s even more potential benefit for operator safety in this setting. “This is going to be a growing area that may even surpass use for coronary interventions. There’s a range of peripheral interventions that appear to be amenable to a robotic approach — below-the-knee, above-the-knee, carotid, renal and mesenteric interventions.”
Advertisement
At this point, the evidence base for robotically assisted interventions has largely been limited to the noncontrolled trials that supported regulatory approval of the first robotic systems, including the PRECISE and CORA-PCI studies in the case of percutaneous coronary intervention (PCI) and the RAPID trial in the case of peripheral vascular intervention (PVI).
Collective evidence from these trials and registry data to date indicate that both short-term outcomes and cardiovascular event rates at one year with robotically assisted PCI and PVI appear to be equivalent to those with manual PCI and PVI. “There has been no signal of a disadvantage in patient safety or other clinical outcomes to date,” Dr. Khatri says, though he points out that no head-to-head comparisons have been conducted.
He adds that a limited body of data suggests that operators can more accurately measure lesion length with the robot, which may allow more reliable deployment of just the right length of stent. “The more precisely you can match stent length to lesion length, the better it is for the patient,” he explains. “So this represents a potential benefit to patients, but it needs to be more thoroughly investigated and confirmed.”
Other potential patient benefits may be on the horizon. One involves “telestenting,” or the use of robotic interventions and telemedicine to broaden access to interventional cardiology care in areas that currently lack it. With appropriate real-time video and audio communication equipment, an experienced operator can perform a case from the remote control cockpit regardless of whether the table is on the other side of a glass wall or hundreds of miles away, so long as the table is equipped with a robotic arm and staffed by an on-site cardiologist or experienced technician. The feasibility of telestenting has already been demonstrated in small published series such as the REMOTE-PCI study.
Advertisement
“Interventions offered remotely via telestenting are not a far-fetched idea and may start to be adopted in the not-too-distant future,” says Dr. Khatri. “This concept can offer real value to patients in rural or remote areas, who will be able to access tertiary center expertise without long-distance travel.”
Another possible benefit that Dr. Khatri foresees is the ability to extract metadata from the robotic devices for development of algorithms to improve the treatment of certain types of lesions.
“I think robotics is going to become the way interventional cardiology is done pretty much everywhere over the next 10 to 15 years,” Dr. Khatri concludes. “It’s going to take some additional development of the technology, but the likely future landscape looks clear.”
He says he’s already near a “robot first” approach in his own practice, as he offers a robot-assisted procedure to virtually all patients whose lesions don’t exclude it because of over-the-wire requirements. He notes that robotic procedure time is “pretty close” to that of manual procedures and that he’s encountered essentially no resistance to robotic procedures from patients, despite the lack of a clear patient benefit at this point.
And he sees real interest in robotics from his fellow interventionalists at Cleveland Clinic and beyond. “Once you demonstrate to your colleagues that this approach is feasible and effective, it’s just a matter of comparing your radiation badge reading to theirs,” he observes. “It quickly becomes a no-brainer.”
Advertisement
Large retrospective analysis may prompt prospective studies
How to talk about lifetime risk, treatment goals, Lp(a) testing, statin skepticism and more
A scannable recap of recent volumes and clinical metrics from Cleveland Clinic
Cleveland Clinic reports first U.S. series focused on use in this challenging setting
Large series confirms early and long-term survival advantages over partial pericardial resection
AVANT GUARD trial extends first-line role for ablation beyond paroxysmal atrial fibrillation
Maintain a high index of clinical suspicion and consider the underlying etiology
Protocol adoption at Cleveland Clinic sharply raised share of transferred patients getting timely PCI