New findings on aortic root position and features may lead to safer valvar surgery
Left bundle branch block (LBBB) or high-grade atrioventricular block, which can necessitate permanent pacemaker implantation, are some of the most serious complications of transcatheter aortic valve replacement (TAVR) and surgeries involving the aortic valve and surrounding area. As the adoption of TAVR has expanded, ways to minimize electrical system-related complications such as LBBB have been the focus of significant research.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
If validated by clinical studies, findings from Cleveland Clinic experts may give surgeons new tools for planning TAVR and aortic valve surgeries that could result in a lower incidence of serious post-procedure conduction system damage. In groundbreaking translational pathologic research, the researchers combined advanced imaging techniques with histology to reveal key features of the conduction axis and relationships between it and the aortic root.
To the best of the authors’ knowledge, theirs is the first study to make inferences linking the vulnerability of the conduction axis below the right coronary sinus to the 3D arrangement of the aortic root.
“Our techniques are novel, in that we were able to pinpoint specific parts of the conduction axis within a millimeter and understand their position relative to the aortic root,” says Justin T. Tretter, MD, first author of the study and Co-Director of the Congenital Valve Procedural Planning Center. “We also took our previous 2D studies further by using 3D imaging to validate an early description of the axis that had been overlooked or underappreciated by other contemporary investigators.”
For the study, the investigators used eight structurally normal donor hearts from adults with no known history of cardiovascular disease, obtained from the National Disease Research Interchange, and two pediatric hearts from children who had undergone autopsy. Their goal was to describe variable features of the aortic root within the ventricular base, which could be used by clinical imagers to inform interventionalists performing TAVR or surgeons performing aortic valve surgery as patient-specific landmarks for vulnerability of the conduction axis.
Advertisement
First, the feasibility of making serial histological sections of the heart was tested on the smaller pediatric organs. Then high-resolution anatomic images of the adult hearts were made with a 3T magnetic resonance (MR) scanner followed by histological sections of these same hearts.
Next, 3D representations of the aortic root in each of the adult hearts were constructed using the MR images to identify variability in the location of the aortic roots within the ventricular base. Histological sections of the organs were simulated from the 3D MR images, and directly compared to the histological sections.
Placement of the simulated histological sections within the reconstructed roots in the ventricular base allowed the researchers to accurately measure the distance between components of the conduction axis and components of the roots. By correlating that information with the location of the roots in the ventricular base, they could infer how vulnerable the conduction axis in a particular heart would be during TAVR.
“Most of what we understand about the conduction system dates back to the early 1900s,” says Dr. Tretter. “Our results confirmed a 1906 description by Tawara that the superior fascicle of the LBB is closely related to the nadir of the right coronary aortic leaflet.” The actual relationship in the adult hearts the authors studied, however, varied.
In three of the eight organs, the overall conduction axis was relatively remote from the aortic valve “annulus” or virtual basal ring, two of which had a centrally positioned aortic root and one that had a significant clockwise rotation. In the other five adult hearts, the conduction axis was much closer to the plane of the virtual base ring throughout its ventricular course. In three of those organs, the root was rotated counterclockwise; in two, it was centrally positioned.
Advertisement
Says Dr. Tretter, “We found four factors in the adult hearts that were predictive of a conduction axis close to the aortic valve: counterclockwise rotational position of the aortic root, a very narrow and shallow inferoseptal recess which positioned the conduction system within the area underneath the aortic root, a membranous septum close to the aortic root, and minimal myocardium to support the right coronary sinus.” Most of those features, he notes, can be visualized on computed tomography, which is often performed prior to TAVR.
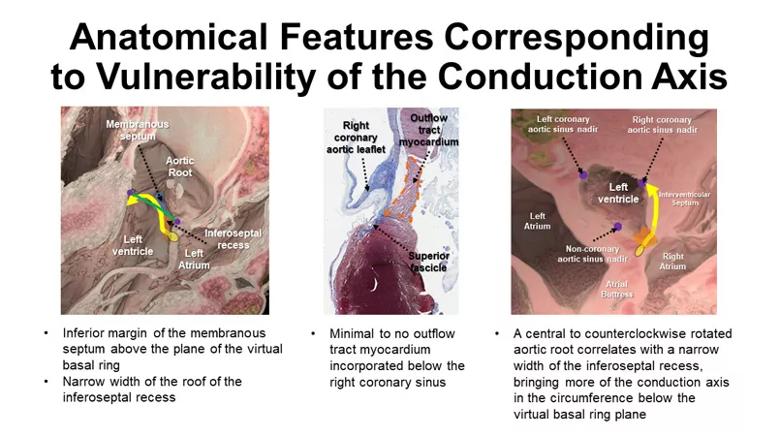
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/5efe6d09-9277-4eb4-89e0-7fd992ae9cdf/23-CHP-3711810-CQD-Tretter-805px_jpg)
Figure. Graphic depicts visual summary of the authors’ findings.
“There have been many attempts to directly image the cardiac conduction system, but there is currently no way to do that on living patients,” says Dr. Tretter. “Our findings, in contrast, are clinically applicable and we are already seeing positive results when applying them in presurgical planning for our patients undergoing surgery for aortic valve and complex left ventricular outflow tract disease. We aim to additionally apply this approach to patients undergoing TAVR.”
Limitations that the investigators noted when describing their study included the small number of hearts used and the fact that they all came from white females, were fixed in a non-hemodynamic state and were anatomically normal organs. The next step in their research is a similar study in hearts from donors with aortic valvar stenosis.
Advertisement
Advertisement

TVT Registry analysis could expand indication to lower surgical risk levels

Patient series and bench validation support efficacy and safety of CLEVE procedure

In the wake of NOTION-3 findings, a strong argument for physician judgment remains
Post hoc analysis of PROTECTED TAVR finds reduced stroke risk in the U.S. but not beyond
Analysis of STS/ACC TVT Registry finds greatest benefit in patients with prior stroke

TAVR explant demands multidisciplinary expertise

How our HVTI Advisory Services team facilitated swift improvements for an allied health organization
Support for a TAVR-first approach in patients with concurrent valve and coronary disease