Randomized WATCH-TAVR trial confirms safety of the combined procedures
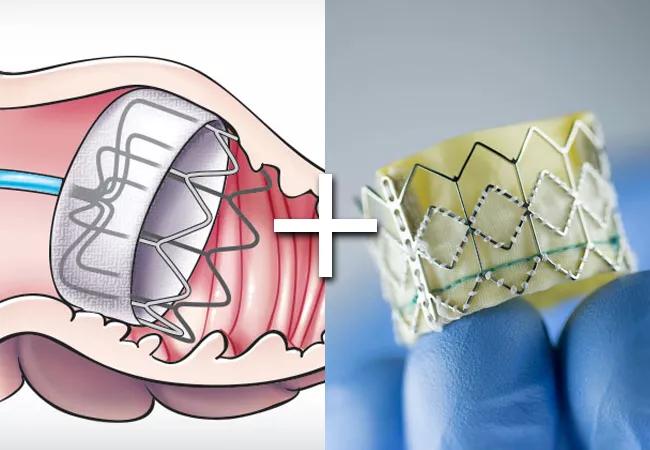
In patients with severe aortic stenosis and atrial fibrillation (AF) undergoing transcatheter aortic valve replacement (TAVR), a strategy of concurrent TAVR and left atrial appendage occlusion (LAAO) with the Watchman™ 2.5 device is noninferior to TAVR plus medical therapy with anticoagulation. So concludes the randomized controlled WATCH-TAVR trial, which was presented by Samir Kapadia, MD, in a late-breaking clinical trials session at the TCT 2023 conference in San Francisco and published simultaneously in Circulation.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“Observational studies have suggested that concomitant TAVR and LAAO is feasible and safe,” says Dr. Kapadia, Chair of Cardiovascular Medicine at Cleveland Clinic. “Now, for the first time, a prospective randomized trial has confirmed that these two procedures can be performed at the same time without an increased risk of complications. These data support LAAO at the time of TAVR as an alternative to chronic oral anticoagulation for the large number of TAVR patients with a history of atrial fibrillation.”
He says the trial is notable for two additional reasons. “This is one of the first studies to evaluate the use of two cardiovascular devices in a single procedure,” he notes, “although additional studies like this are in the works. This type of combined procedural approach offers potential benefits in terms of patient convenience, enhanced safety through avoidance of multiple procedures and potential overall cost efficiencies. Moreover, the study is novel in that it allowed patients in the medical therapy arm to receive whatever anticoagulation therapy their physician deemed appropriate, whether warfarin or a direct oral anticoagulant (DOAC). The trials that led to Watchman’s FDA approval showed noninferiority to warfarin before DOACs were widely used, so WATCH-TAVR adds new insights on the device’s merits relative to contemporary anticoagulation strategies.”
The trial was prompted by the prevalence of AF among TAVR patients, which has been reported to range from 15% to 40%. Moreover, the presence of AF confers an increased risk of death, stroke and rehospitalization among patients undergoing TAVR.
Advertisement
“It’s notable that the major TAVR trials have shown a stroke rate of 2% to 6% during the period from one month to one year after the procedure, which makes stroke beyond the periprocedural period an outcome of interest,” Dr. Kapadia says. “This, along with the many shared comorbidities between aortic stenosis and AF, and high rates of major bleeding in the year after TAVR — which may be due in part to anticoagulation required for AF — compelled us to undertake this investigation.”
The study was conducted as a prospective open-label investigation at 34 North American sites from late 2017 to late 2020. Patients with planned TAVR and documented AF who were tolerant of oral anticoagulants were eligible.
All patients in the TAVR+LAAO group received the Watchman 2.5 device with adjunctive pharmacotherapy based on Watchman 2.5 labeling at the time: 45 days of warfarin and aspirin followed by 4.5 months of dual antiplatelet therapy. Patients in the TAVR+medical therapy group were treated with anticoagulation — either warfarin or a DOAC — with or without antiplatelet therapy, with the choice of specific regimen at the discretion of their physician.
The primary endpoint for noninferiority between the two strategies was first occurrence of a composite of all-cause mortality, stroke or major bleeding at two years after TAVR. Various outcomes were assessed as secondary endpoints.
Of the study’s 349 patients, 177 were randomized to TAVR+LAAO and 172 to TAVR+medical therapy. The overall cohort had a mean age of 81 years, a mean CHA2DS2-VASc score of 4.9 and a mean HAS-BLED score of 3.0. At baseline, 85.4% were taking anticoagulation and 71.3% were taking antiplatelet therapy. Baseline characteristics were well matched between the two treatment cohorts.
Advertisement
Clinical outcomes. Rates of the composite primary endpoint were 22.7 events/100 patient-years in the TAVR+LAAO arm versus 27.3 events/100 patient-years in the TAVR+medical therapy arm, which met the predefined criteria for noninferiority (P < 0.001 for noninferiority) but did not demonstrate statistical superiority of TAVR+LAAO.
Noninferiority was maintained in both intention-to-treat and as-treated analyses and across prespecified patient subgroups defined by gender, age, presence of diabetes, history of stroke and type of AF. Moreover, rates of the individual components of the primary endpoint (all-cause death, stroke, major bleeding) were similar between the groups at two years of follow-up.
Rates of major bleeding were numerically higher in the TAVR+LAAO arm during the initial six months but were numerically lower in this arm between six months and two years. “The initial increase in bleeding with LAAO was likely attributable to the study protocol’s mandated use of warfarin plus aspirin for 45 days followed by four and a half months of dual antiplatelet therapy, based on the approved label for Watchman 2.5 at the time,” Dr. Kapadia notes. “This labeling has subsequently been updated to permit use of DOACs or dual antiplatelet therapy with the latest-generation Watchman FLX device. It’s likely that less-intensive anticoagulation and antiplatelet regimens like those being studied in the ongoing CHAMPION-AF trial of the Watchman FLX may be associated with reduced bleeding after device implantation, although we await those data.”
Advertisement
The only adjudicated outcome that was significantly different between the groups at two years was arterial or venous thrombosis or embolism, which was higher in the TAVR+LAAO arm (5.5 vs. 1.1 events/100 patient-years), driven primarily by venous thrombosis.
Procedural details and post-procedure medications. As expected, procedural duration was greater in the TAVR+LAAO arm (by a median of 38 minutes), as was contrast volume (median of 119 mL vs. 70 mL); use of general anesthesia and transesophageal echocardiography (TEE) was more frequent with the combined procedures. “This latter procedural limitation may be minimized, however, by the fact that LAAO is increasingly performed without general anesthesia through the use of intracardiac echo guidance in lieu of TEE,” Dr. Kapadia observes. Length of stay did not differ between the treatment groups.
At two years, the share of patients on any antiplatelet therapy was 82.5% in the TAVR+LAAO arm versus 50.8% in the TAVR+medical therapy arm; the share of patients on any anticoagulation therapy was 13.9% in the TAVR+LAAO arms versus 66.7% in the TAVR+medical therapy arm. “It’s notable that only two-thirds of patients who underwent TAVR alone remained on anticoagulation at two-year follow-up,” Dr. Kapadia says. “This raises concern about the continued long-term risk of stroke in patients not on anticoagulation and underscores the singular opportunity offered by LAAO to provide long-term stroke protection.”
In view of these findings of noninferiority in a well-designed controlled trial, the WATCH-TAVR investigators call for reimbursement of concurrent TAVR and LAAO for appropriate TAVR patients with nonvalvular AF.
Advertisement
“We are reaching out to the Centers for Medicare & Medicaid Services (CMS) to encourage reimbursement of the combined procedures,” Dr. Kapadia says. “From the standpoint of patient convenience, as well as safety, cost and avoidance of repeat interventions, performing TAVR and LAAO as concomitant procedures can make a lot of sense from a value perspective.”
“This trial is really important for patients with aortic stenosis and atrial fibrillation,” adds study co-author Amar Krishnaswamy, MD, Section Head of Invasive and Interventional Cardiology at Cleveland Clinic. “I hope the FDA and CMS will see this patient-centric trial as evidence for appropriate reimbursement of the combined procedure so that patients don’t have to return for their Watchman as a second procedure after TAVR. Unfortunately, the expense of the device will not allow any providers to place it as part of a combined procedure unless it is covered appropriately.”
The study was coordinated by the Cleveland Clinic Coordinating Center for Clinical Research (C5Research) and funded as an independent study by Boston Scientific, which markets the Watchman device.
Advertisement

TVT Registry analysis could expand indication to lower surgical risk levels

Patient series and bench validation support efficacy and safety of CLEVE procedure

In the wake of NOTION-3 findings, a strong argument for physician judgment remains
Post hoc analysis of PROTECTED TAVR finds reduced stroke risk in the U.S. but not beyond
Analysis of STS/ACC TVT Registry finds greatest benefit in patients with prior stroke

TAVR explant demands multidisciplinary expertise

How our HVTI Advisory Services team facilitated swift improvements for an allied health organization
Support for a TAVR-first approach in patients with concurrent valve and coronary disease