Leading-edge technology will improve diagnosis and prognosis
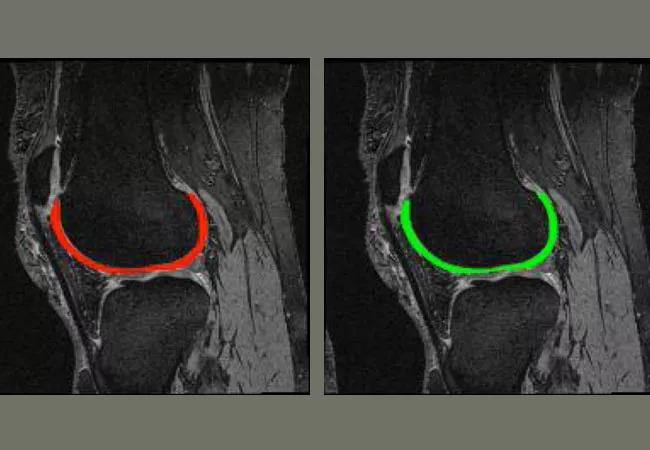
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/ef1b4574-cd79-46bc-a937-b5c2d02bbeb9/18-ORT-1303-cartilageSegment-_650x450_jpg)
18-ORT-1303-cartilageSegment-_650x450
By Xiaojuan Li, PhD; Carl Winalski, MD; and Kurt Spindler, MD
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Osteoarthritis (OA) affects more than 27 million people in the United States and has been recognized as one of the fastest growing medical conditions worldwide. Ongoing efforts for novel pharmacological therapies continue but there are no disease-modifying OA drugs (DMOADs) to slow or halt cartilage degeneration.
One of the hurdles in developing DMOADs is the lack of sensitive and reliable noninvasive biomarkers to efficiently detect treatment effects. Such biomarkers would also help identify patients at risk for developing OA or at an early stage of disease when prevention strategies, such as weight loss, diet changes and physical exercise, are shown to be most effective in slowing down disease progression.
A focus of the newly formed Program of Advanced Musculoskeletal Imaging (PAMI) is to develop novel quantitative MRI techniques for improving early diagnosis and prognosis for OA and post-traumatic OA (PTOA).
We have developed 3D quantitative MR T1ρ and T2 imaging techniques that can detect biochemical changes within the collagen-proteoglycan matrix of articular cartilage at early stages of degeneration and have observed significant correlations between MR T1ρ measures with proteoglycan concentrations, and biomechanical and histological evaluation of specimens harvested from patients who underwent total knee arthroplasty. MR T1ρ values also were elevated in lesions confirmed with arthroscopic evaluations in patients with acute knee injuries such as anterior cruciate ligament (ACL) tears. In patient cohort studies, we observed significantly higher T1ρ and T2 values in patients with OA and knee injuries as compared to healthy controls. Most interestingly, baseline cartilage T1ρ and T2 has shown to predict cartilage morphologic lesion development and patient-reported outcomes (such as pain) at 1-, 2- and 3-years after ACL reconstruction. These results suggest that T1ρ and T2 may serve as promising biomarkers to improve early diagnosis and prognosis of cartilage health and OA.
Advertisement
Despite promising results, challenges remain, including lack of standardized acquisition and quantification methods, and long acquisition times. Multicenter trials using cartilage T1ρ and T2 imaging have been primarily limited to single vendor MR systems thereby eliminating pulse sequence and RF coil differences between manufacturers. To address these issues, we have performed a multi-center multi-vendor cross-validation study of cartilage T1ρ and T2 imaging, sponsored by the Arthritis Foundation. Outcomes will pave the way toward large-scale multi-vendor, multi-site trials for OA using advanced quantitative MRI.
In addition, we have developed an atlas based voxel-based relaxometry analysis (VBR) that will provide evaluation of very focal changes that will be otherwise masked by conventional region of interest (ROI) based methods. We also have been working on developing fast imaging and automatic analysis techniques to facilitate clinical translation of cartilage T1ρ and T2 imaging. Using MR-compressed sensing and parallel imaging techniques, adaptive to cartilage signal, we have shown acquisition time can be shortened four times without sacrificing image quality and quantification accuracy (with T1ρ and T2 quantification error < 1%).
We also are developing deep learning techniques for fully automatic cartilage segmentation and have achieved 89 percent accuracy by combining a Conditional Generative Adversarial Network (cGAN) and a Convolutional Neural Network (CNN, U-NET), which outperformed state-of-the-art automatic cartilage segmentation using deep learning techniques. Combining fast imaging techniques with automatic tissue segmentation and quantification will significantly advance efforts toward clinical translation of quantitative cartilage MRI techniques.
Advertisement
Finally, we are optimizing the T1ρ and T2 sequences on research-dedicated MR scanners, and expect to soon add fast and reliable compositional imaging sequences to routine clinical protocols on clinical scanners, which will allow us to correlate clinical outcomes with compositional maps acquired prospectively as part of routine care.
Dr. Li is the Director of the Program for Advanced Medical Imaging in the Department of Biomedical Engineering at Cleveland Clinic Lerner Research Institute. Dr. Winalski is a diagnostic radiologist and the Clinical Director of the Program for Advanced Medical Imaging. Dr. Spindler is an orthopaedic surgeon and sports medicine expert at Cleveland Clinic.
Advertisement
Advertisement
Study highlights the need for objective functional measures as value-based care expands
Reduces likelihood of achieving CMS-defined substantial clinical benefit at one year
Step counts suggest recovery is more variable and individualized than commonly believed
Innovations and practical insights for safe return to sport after lower-limb injury
Physical therapy before surgery is intended to optimize outcomes after surgery
Recognition could help guide health policy, workforce development and resource allocation worldwide
The rare and aggressive form of bone cancer requires specialized treatment, but new and emerging therapies are providing hope for patients
With a better understanding of timelines and contributing factors, clinicians can help athletes undergoing TKAs set realistic expectations