Viabahn VBX offers a safe alternative option for endovascular aneurysm repair
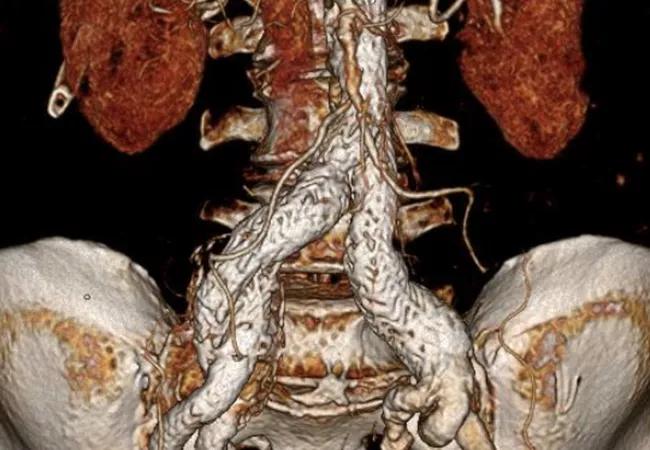
Image content: This image is available to view online.
View image online (https://assets.clevelandclinic.org/transform/31477fca-00e6-411a-9016-b4bb8de85678/22-HVI-2928208-CQD-650x450-1_jpg)
22-HVI-2928208 CQD 650×450
The off-label addition of the Gore Viabahn VBX (VBX) balloon expandable endoprosthesis to the Gore Excluder iliac branch endoprosthesis (IBE) is a comparable alternative to the FDA-approved internal iliac component (IIC) added to the IBE for treatment of aortoiliac artery aneurysms. So found a retrospective single-center review of patients treated with either procedural approach, which resulted in similar safety and efficacy outcomes while allowing for more sizing options and additional access points with the Viabahn VBX. The study was recently published online ahead of print in the Journal of Vascular Surgery.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“Our findings suggest that the VBX is an equal substitute for the internal iliac component of the Excluder iliac branch endoprosthesis while allowing more flexibility in treating a variety of challenging anatomies,” says study co-author Francis Caputo, MD, of Cleveland Clinic’s Department of Vascular Surgery. “This is good news for expanding IBE use to more patients.”
The IBE is the only iliac branch device currently approved in the U.S. for preserving blood flow to both the external and internal iliac arteries.
The self-expanding IIC was designed to be used with the IBE for internal iliac artery reperfusion, but the separate VBX covered stent — which is FDA-approved for treating arterial occlusive disease in the setting of de novo or restenotic lesions in the common iliac artery and/or external iliac artery — is increasingly used instead because it offers more treatment diameters, lengths and delivery options. However, data are scarce on use of the VBX in this off-label setting.
The current study was designed to retrospectively assess outcomes for patients receiving the IBE combined with either the IIC or the VBX stent.
Patients who were treated at Cleveland Clinic for aortoiliac artery aneurysms with the IBE plus either the IIC or VBX between February 2016 and March 2021 were included. The study population consisted of 62 patients with 64 treated arteries, with the IIC used for 35 arteries and the VBX for 29.
Mean follow-up was 24.1 ± 14.6 months; the follow-up period for the IIC was significantly longer than that of the VBX (29.9 ± 13.9 month vs. 17.0 ± 12.3 months, P < 0.001) because the VBX only became available later in the study period.
Advertisement
A variety of clinical outcomes were assessed at regular intervals. No significant differences were found in any of the outcomes of interest, including technical success, presence of endoleak on completion, readmission, 30-day mortality, requirement for any IBE reintervention after 30 days, or internal iliac limb primary patency. No return to the operating room was required, and no type Ia, Ib or III endoleaks occurred in either group.
The study indicates that use of the VBX stent with the IBE in the treatment of aortoiliac aneurysms results in similar outcomes as the FDA-approved IIC with the IBE. As the VBX offers greater device variety, Dr. Caputo notes that the VBX could allow for treatment in situations other than what is currently included in the IBE’s instructions for use.
For example, the authors write that if a repair must be extended beyond the internal iliac artery bifurcation with more length needed, the VBX stent “can be tapered to avoid diameter discrepancies and poor apposition to the vessel wall.” They add that the angle of approach necessary to cannulate the internal iliac artery before stenting sometimes cannot be achieved with contralateral femoral access and can be technically challenging using ipsilateral access. “In such cases,” Dr. Caputo notes, “upper extremity arterial access and placement of the longer-delivery VBX is a viable and safe alternative.”
He adds that pooled data from multiple institutions, especially if collected in a prospective, randomized fashion, would likely better define which patients might benefit most from this alternative approach of VBX use with IBE. Longer-term observations and prospective comparative studies in matching cohorts could also further clarify its use. In the meantime, he notes that this study provides important evidence for expanding treatment options for internal iliac artery preservation.
Advertisement
“We have found that the VBX stent allows landing in the anterior or posterior division branches of the internal iliac artery, which requires considerable diameter tapering from the IBE gate,” adds study co-author Sean Lyden, MD, Chair of Vascular Surgery at Cleveland Clinic. “Our experience indicates that IBE use with the VBX stent instead of the IIC is safe and effective for aortoiliac artery aneurysm exclusion with preservation of internal iliac artery flow.”
The authors attested that the manufacturer of the devices in this study, W.L. Gore & Associates, had no role in the study.
Advertisement
Advertisement
Large retrospective analysis may prompt prospective studies
How to talk about lifetime risk, treatment goals, Lp(a) testing, statin skepticism and more
A scannable recap of recent volumes and clinical metrics from Cleveland Clinic
Cleveland Clinic reports first U.S. series focused on use in this challenging setting
Large series confirms early and long-term survival advantages over partial pericardial resection
AVANT GUARD trial extends first-line role for ablation beyond paroxysmal atrial fibrillation
Maintain a high index of clinical suspicion and consider the underlying etiology
Protocol adoption at Cleveland Clinic sharply raised share of transferred patients getting timely PCI