Tag debug info: client: {"assets":{},"datasets":{},"live":{},"projects":{},"users":{},"observable":{"assets":{},"datasets":{},"live":{},"projects":{},"users":{}}} Now: 1770745636420 Cache Key: cqdTagPageBySlug:glioblastoma fetchCache[cqdTagPageBySlug:glioblastoma].expirationTime: falsey fetchCache[cqdTagPageBySlug:glioblastoma]. seconds remaining: falsey All fetchCache expiration times: -- Key: cqdNotFoundPage, seconds remaining: 975 -- Key: cqdTagPageBySlug:emergency-room-er, seconds remaining: -126095 -- Key: cqdPostsByTag:cqd-migrated-tag-4246,1,10, seconds remaining: -126009 -- Key: cqdTagPageBySlug:localized-kidney-cancer, seconds remaining: -10738 -- Key: cqdPostsByTag:cqd-migrated-tag-26497,1,10, seconds remaining: -10649 -- Key: cqdTagPageBySlug:cardiometabolic-disease, seconds remaining: -9831 -- Key: cqdPostsByTag:cqd-migrated-tag-21506,1,10, seconds remaining: -9749 -- Key: cqdTagPageBySlug:samantha-anne-md, seconds remaining: -8934 -- Key: cqdPostsByTag:cqd-migrated-tag-7607,1,10, seconds remaining: -8855 -- Key: cqdTagPageBySlug:volatile-organic-compounds-voc, seconds remaining: -8038 -- Key: cqdPostsByTag:cqd-migrated-tag-17359,1,10, seconds remaining: -7966 -- Key: cqdTagPageBySlug:patient-experience-summit, seconds remaining: -6685 -- Key: cqdPostsByTag:cqd-migrated-tag-1955,2,10, seconds remaining: -6610 -- Key: cqdTagPageBySlug:nonprescription-statins, seconds remaining: 975 -- Key: cqdPostsByTag:cqd-migrated-tag-23741,1,10, seconds remaining: 1050 -- Key: cqdTagPageBySlug:specialty-infusion-nurse, seconds remaining: 1424 -- Key: cqdPostsByTag:cqd-migrated-tag-22639,1,10, seconds remaining: 1502 -- Key: cqdTagPageBySlug:c5research, seconds remaining: 1846 -- Key: cqdPostsByTag:cqd-migrated-tag-1844,1,10, seconds remaining: 1926 -- Key: cqdTagPageBySlug:transient-receptor-potential-vanilloid-4-trpv4, seconds remaining: 3228 -- Key: cqdPostsByTag:cqd-migrated-tag-1715,1,10, seconds remaining: 3304 -- Key: cqdTagPageBySlug:androgen-receptor-ar-antagonists, seconds remaining: 5943 -- Key: cqdPostsByTag:cqd-migrated-tag-22696,1,10, seconds remaining: 6016 -- Key: cqdTagPageBySlug:covid-19-safety-precautions, seconds remaining: 6391 -- Key: cqdPostsByTag:cqd-migrated-tag-22361,1,10, seconds remaining: 6471 -- Key: cqdTagPageBySlug:immune-mediated-inflammatory-diseases-imids, seconds remaining: 8643 -- Key: cqdPostsByTag:cqd-migrated-tag-9523,1,10, seconds remaining: 8719 conditions: -- false, -- NA, -- NA, -- NA -- false Cache miss for key cqdTagPageBySlug:glioblastoma - retrieving from Sanity CCCache.dataFetchCount: 599 Cache cleanup seconds remaining: 9262
Advertisement
Advertisement
Advances in genomics, spinal fluid analysis, wearable-based patient monitoring and more
Researchers use AI tools to compare clinical events with continuous patient monitoring
Cleveland Clinic researchers pursue answers on basic science and clinical fronts
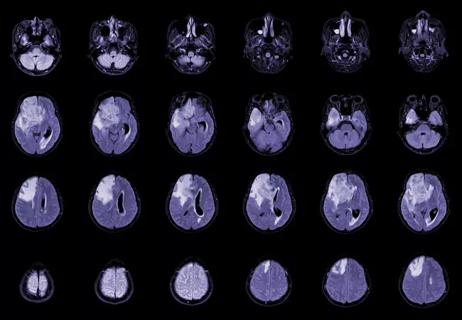
New research from Cleveland Clinic helps explain why these tumors are so refractory to treatment, and suggests new therapeutic avenues
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
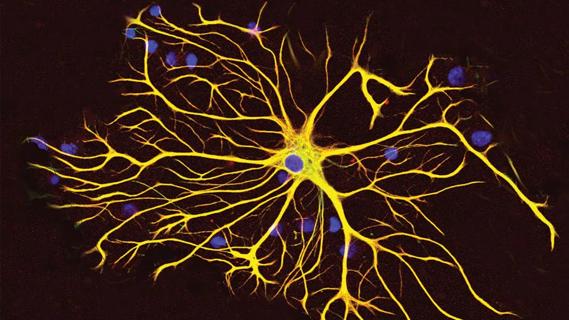
New research focuses on tumorigenic aspects of communication among brain cells
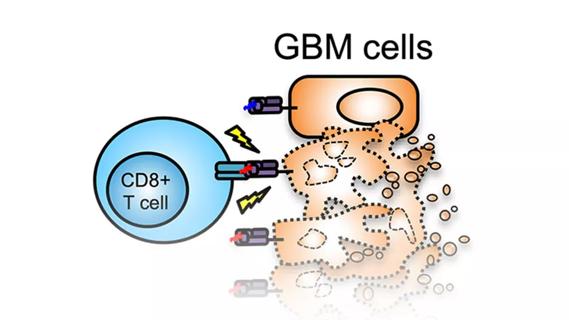
Study demonstrates its role in tumor lethality, raises prospect of therapeutic targets
Can T-cell immunophenotyping help inform treatment decisions?
Intracranial injection of supercharged immune cells proceeds to phase 2 testing
BACE1 inhibitor reduces cancer progression in models of glioblastoma
Findings implicate hydrogen sulfide dysfunction, point to potential treatment strategies
Rendered: Tue Feb 10 2026 17:47:16 GMT+0000 (Coordinated Universal Time)
9500 Euclid Avenue, Cleveland, Ohio 44195 |
800.223.2273 | ©
2026 Cleveland Clinic. All Rights Reserved.