TEER is found to be a safe and good option for severe TR in select patients
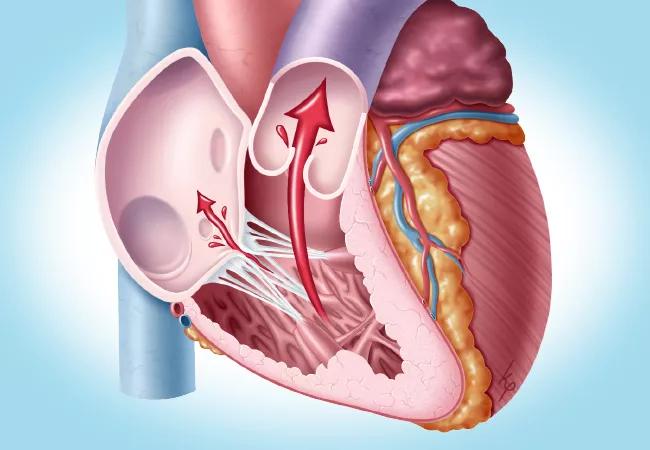
Recently released one-year findings from the TRILUMINATE Pivotal trial are encouraging: tricuspid transcatheter edge-to-edge repair (TEER) using the TriClip Transcatheter Tricuspid Valve Repair System for isolated severe tricuspid regurgitation (TR) safely reduces TR with concomitant improvement in quality-of-life measures, comparing favorably to medical therapy. Results of the prospective randomized trial, conducted in 350 patients at 65 centers in North America and Europe, were reported in a late-breaking clinical trial presentation at the American College of Cardiology’s 2023 scientific session and simultaneously published online in the New England Journal of Medicine.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
“There are few good therapy options for severe isolated TR, so being able to offer a safe and effective nonsurgical approach is very consequential,” says Samir Kapadia, MD, Chair of Cardiovascular Medicine at Cleveland Clinic and principal investigator of the Cleveland Clinic TRILUMINATE trial site. “Based on the trial results, I expect the FDA may approve this therapy for select patients.”
Untreated severe TR is associated with considerable morbidity and reduced survival. Most patients with isolated TR are poor candidates for surgical valve repair or replacement because of comorbidities. In general, surgery for TR is recommended only for those who are undergoing left heart cardiac surgery. Medical therapy — consisting of diuretics — reduces symptoms in some patients.
TEER involves percutaneous deployment of a clip to hold the tricuspid valve leaflets together and thereby reduce regurgitant flow. The Triluminate Pivotal trial randomly assigned patients seen between 2019 and 2021 with symptomatic severe TR to either TEER or medical therapy, with 175 patients in each group. Mean age was 78 years, and 55% were women.
Results included a win ratio at one year of 1.48 (95% CI, 1.06-2.13; P = 0.02) in favor of TEER for the primary end point — a hierarchical composite of death or tricuspid valve surgery; hospitalization for heart failure; and improvement in quality of life, measured with the Kansas City Cardiomyopathy Questionnaire (KCCQ).
TEER was also effective in reducing TR: at 30 days, TR severity was moderate or less in 87.0% of TEER patients versus only 4.8% of the medical therapy group (P < 0.001). The decrease in TR severity in the TEER group appeared stable at one year.
Advertisement
In addition, KCCQ quality-of-life scores at one year increased by a mean of 12.3 points in the TEER group versus 0.6 points in the medical therapy group (P < 0.001). The incidence of death or tricuspid valve surgery was not significantly different between the two groups, nor was the rate of hospitalization for heart failure.
The trial will continue to follow patients for five years.
“TRILUMINATE found TEER to be safe, with reduction of TR correlating with improved quality of life,” summarizes Dr. Kapadia. He cautions, however, that the trial was open-label and that TEER is not applicable to everyone who is a poor candidate for surgery for severe symptomatic TR. Additionally, if the TriClip fails, a patient’s future options may be reduced. “Can you replace the valve with the clip in place?” Dr. Kapadia asks. “It has been done, but it’s not standard procedure.”
He notes that the trial had many exclusion criteria, including high pulmonary artery pressure; severe uncontrolled hypertension; a prior tricuspid valve procedure, pacemaker or ICD leads that would prevent proper device placement; and tricuspid valve leaflet anatomy that was not evaluable or could preclude clip implantation, such as severe coaptation defect or calcification.
“TRILUMINATE used a very select population, and only about 10% to 20% of our population at Cleveland Clinic with severe symptomatic TR qualified to participate,” Dr. Kapadia says.
The authors of the study report comment that death and hospitalization for heart failure occurred less frequently than predicted in both groups, likely related to the strict selection criteria that excluded many of the sickest patients. It is possible that the finding of no significant difference in death and heart failure hospitalization rate at one year between the groups could be attributed to this issue.
Advertisement
Dr. Kapadia identifies the study’s open-label design as an important criticism. “When patients who receive clips are asked to evaluate their quality of life, there can be a bias toward perceived improvement among these patients compared with those in the control group, who are unlikely to feel better since no additional therapy was offered to them,” he says. “Since death and heart failure admissions were not numerically lower compared with controls, the KCCQ data may need additional scrutiny when we interpret the trial’s results.”
“Optimal visualization of the tricuspid valve is critical for the success of these procedures,” says Serge Harb, MD, a multimodality cardiac imager involved in tricuspid TEER procedures at Cleveland Clinic. “This requires a comprehensive understanding and proficient use of advanced multimodality 3D imaging. Imaging plays an indispensable role in every step of the process, from patient selection (identifying those who are likely to benefit) to procedural planning (evaluating feasibility of the procedure and determining the optimal treatment strategy) and interventional guidance (with the aid of real-time 3D multiplanar reconstruction). Imaging is also essential for follow-up evaluation, enabling assessment of the procedure’s efficacy and detection of potential complications. For these reasons, use of advanced multimodality 3D imaging is vital for successful tricuspid valve procedures, which may ultimately lead to improved patient outcomes.”
Dr. Kapadia notes that patients in TRILUMINATE were sometimes disqualified because their valve could not be well visualized with transesophageal echocardiography; however, the more recent use of intracardiac echocardiography in such cases has now lessened that issue and would likely allow more patients to qualify.
Advertisement
Percutaneous replacement of the tricuspid valve is an additional important therapy for TR, Dr. Kapadia points out. This treatment option is being actively investigated in another multicenter trial, TRISCEND, that has finished enrollment and will have results available at the beginning of 2024.
Another emerging option for severe TR is the TricValve® Transcatheter Bicaval Valves System, in which two self-expanding biological valves are percutaneously placed in the superior and inferior venae cavae. It is designed to ameliorate TR without directly addressing the tricuspid valve. (See details in a previous Consult QD post.) Dr. Kapadia and Cleveland Clinic interventional cardiologist Rishi Puri, MD, PhD, are principal investigators of the multicenter randomized TRICAV study comparing TricValve therapy with guideline-directed medical therapy.
Other transcatheter devices designed to address isolated TR are also being studied. “We can expect that more options will continue to emerge,” Dr. Kapadia notes.
“While the TRILUMINATE results are a promising and important first step, we look forward to novel treatment options that can be used in the future in high-risk surgical patients who did not meet the screening criteria for this study,” adds Cleveland Clinic cardiothoracic surgeon James Yun, MD, PhD.
His colleague Amar Krishnaswamy, MD, concurs. “It is very satisfying when patients return to clinic after a successful tricuspid valve intervention and are thankful for how much better they feel and their quality-of-life improvements,” says Dr. Krishnaswamy, Section Head of Invasive and Interventional Cardiology. “As we are in the early phase of understanding these percutaneous therapies for TR, I look forward to the options we can bring to patients in the years ahead.”
Advertisement
Advertisement

Study supports addressing mitral regurgitation before mild tricuspid regurgitation progresses

TRISCEND II trial reports 1-year results

Limited data and experience will translate to a cautious rollout
Cleveland Clinic study argues against waiting for symptoms to develop
Understanding VC anatomy is critical for transcatheter tricuspid valve interventions

How Cleveland Clinic is using and testing TMVR systems and approaches

NIH-funded comparative trial will complete enrollment soon

How Cleveland Clinic is helping shape the evolution of M-TEER for secondary and primary MR